More well thought out work can be found at — https://axial.substack.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you — email us at info@axialvc.com
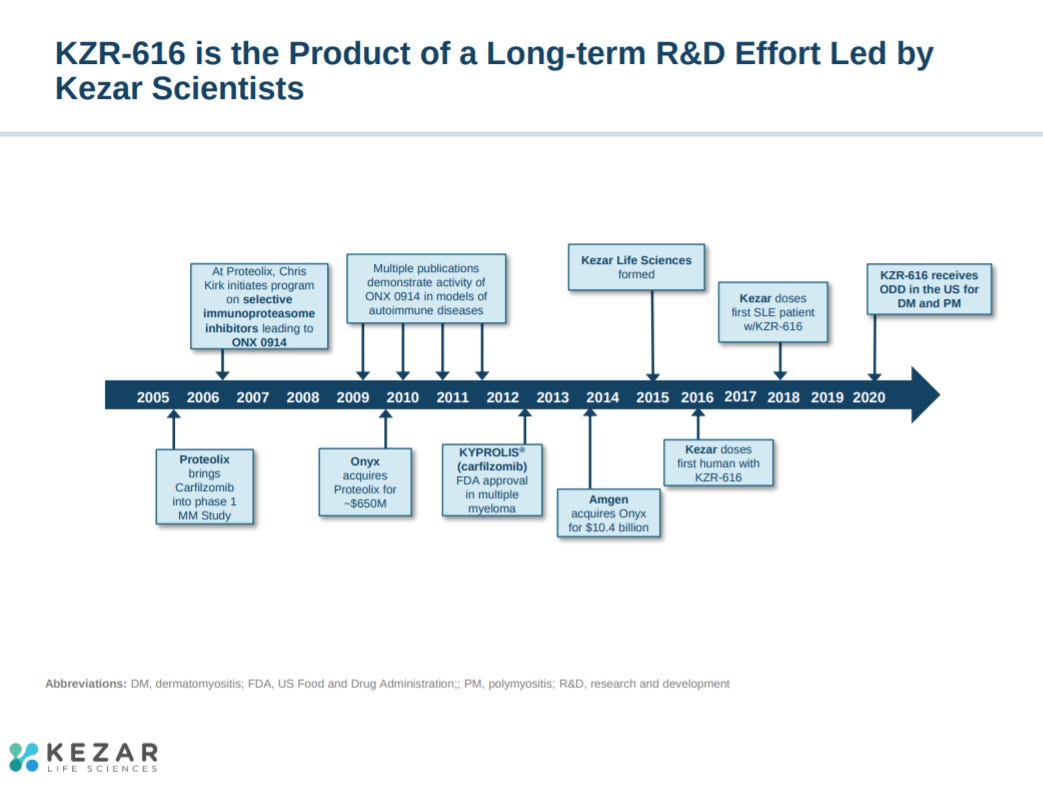
Kezar Life Sciences was founded in 2015 by John Fowler, Jack Taunton, and Christopher Kirk to drug the immunoproteasome for autoimmunity. The company was centered around a licensing agreement with Amgen (from its Onyx/Proteolix acquisition) for IP on an immunoproteasome program. Kirk had been a director at Proteolix and Onyx, after Onyx’s acquisition of Proteolix, where he helped develop Kyprolis (a proteasome inhibitor) to approval in multiple myeloma. Actually, the asset Kezar licensed from Amgen in 2015 actually was from Kirk’s work that had begun in 2006. The company is still in early clinical development, but the ability to drug the immunoproteasome (selective for immune cells) has similar potential as Kyprolis and other proteasome inhibitors. The immunoproteasome is essential to process viral proteins into antigens as well as activate macrophages, regulate cytokine expression, and differentiate subsets of T-cells. As a result, Kezar has a drug candidate that could become a pipeline-in-a-pill for autoimmunity.
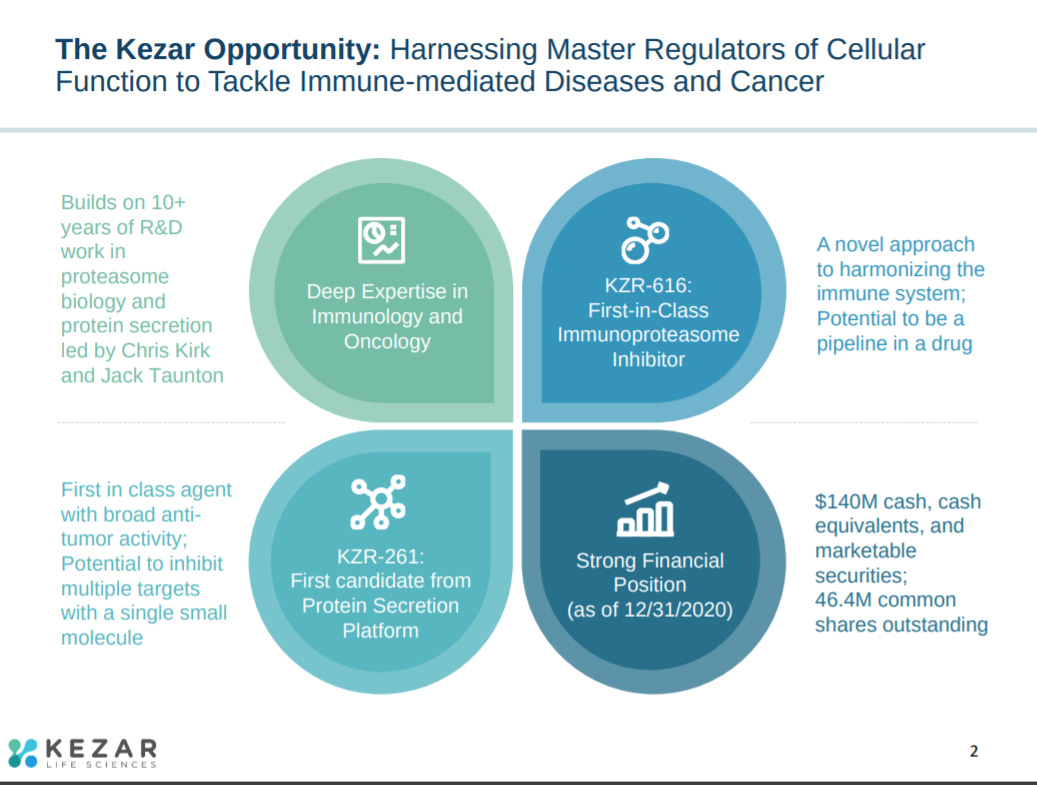
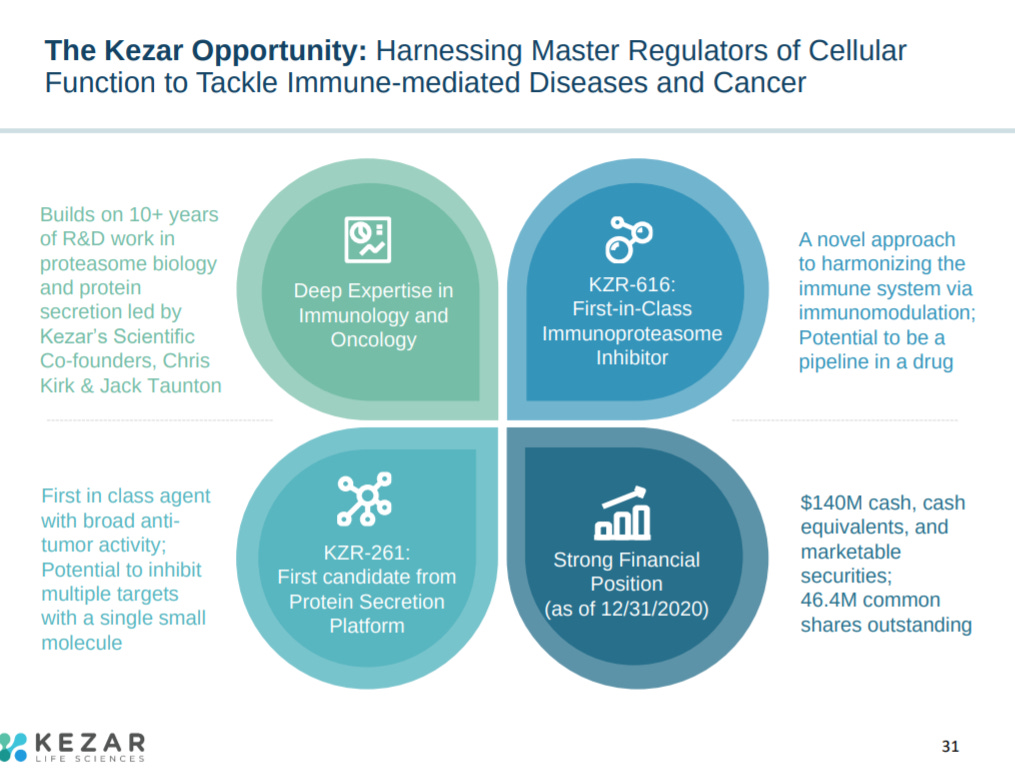
The first slide of their latest corporate presentation provides 4 highlights for Kezar: (1) expertise in the proteasome, (2) first-in-class drug candidate, (3) second drug asset focused on protein secretion, and (4) enough cash to run a phase 2 trial for its lead asset.
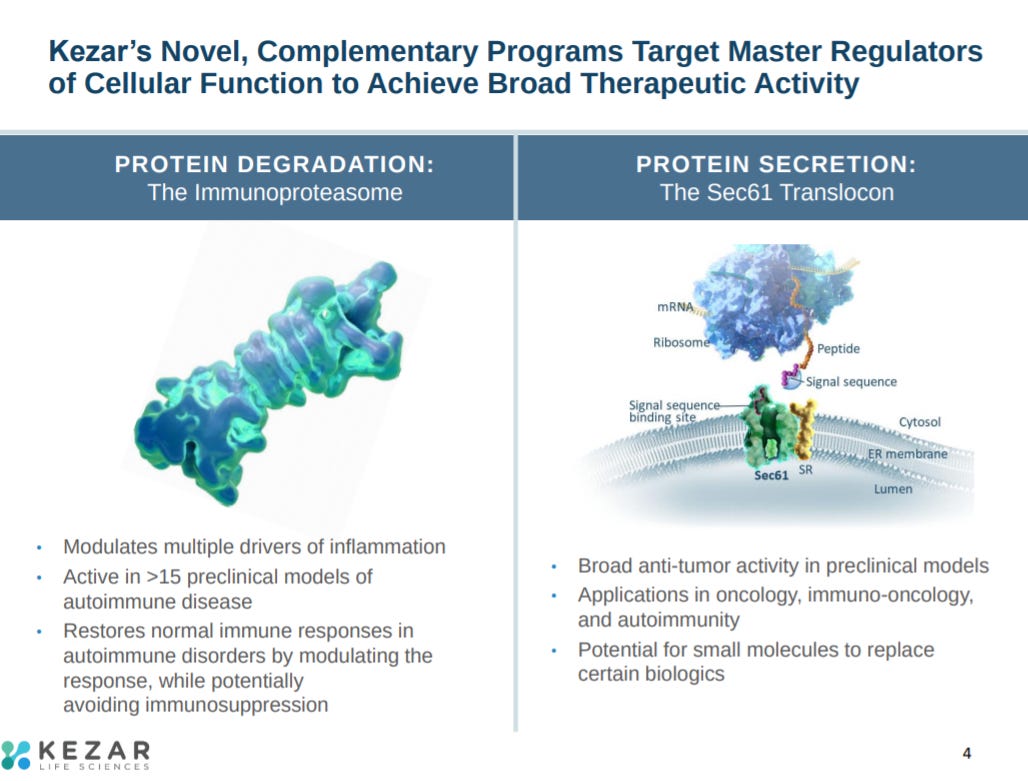
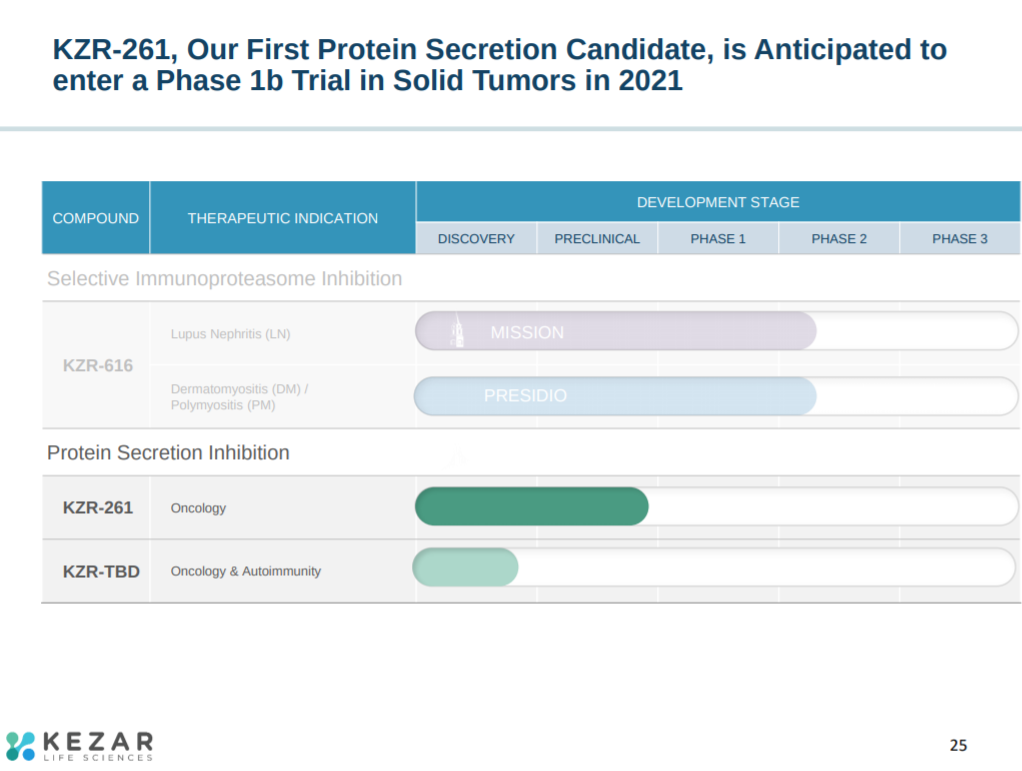
The next slide explains Kezar’s pipeline. The company’s lead asset, KZR-616, is a selective immunoproteasome inhibitor in phase 2 clinical trials for lupus nephritis, dermatomyositis, and polymyositis. When the Kezar was founded, they also initiated programs for protein secretion inhibition - the company has 2 pre-clinical programs in oncology.
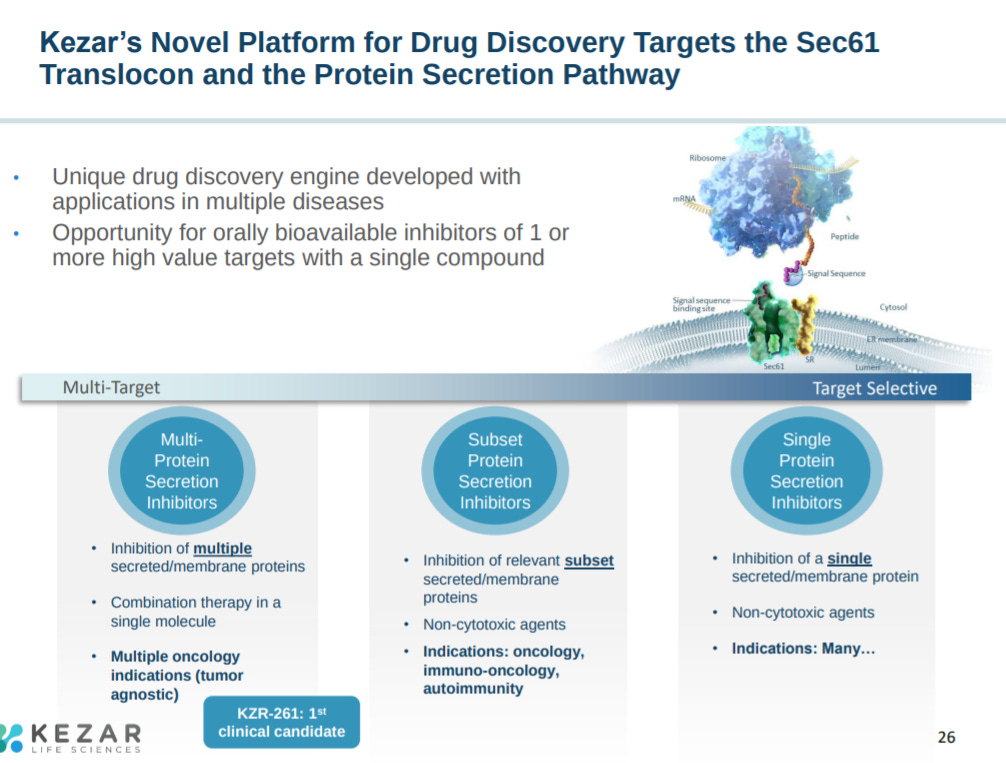
The next slide explains the company’s focus on the immunoproteasome and Sec61 (mediates protein translocation across the endoplasmic reticulum).
Kezar adds a history timeline slide.
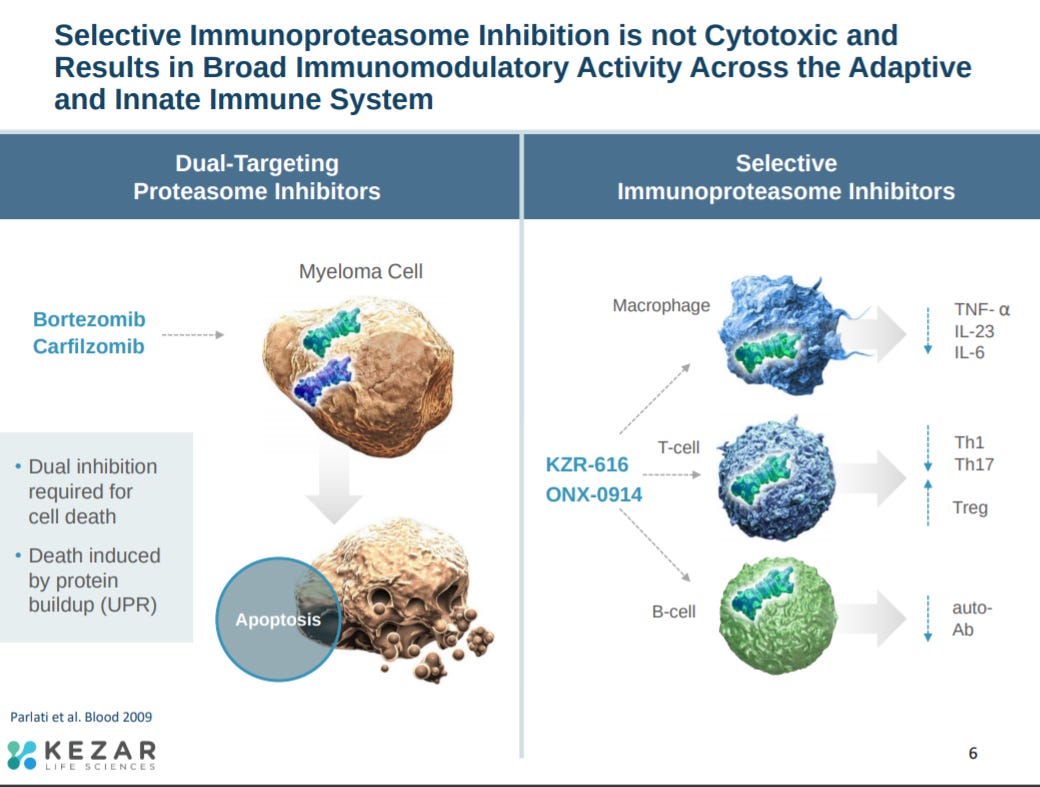
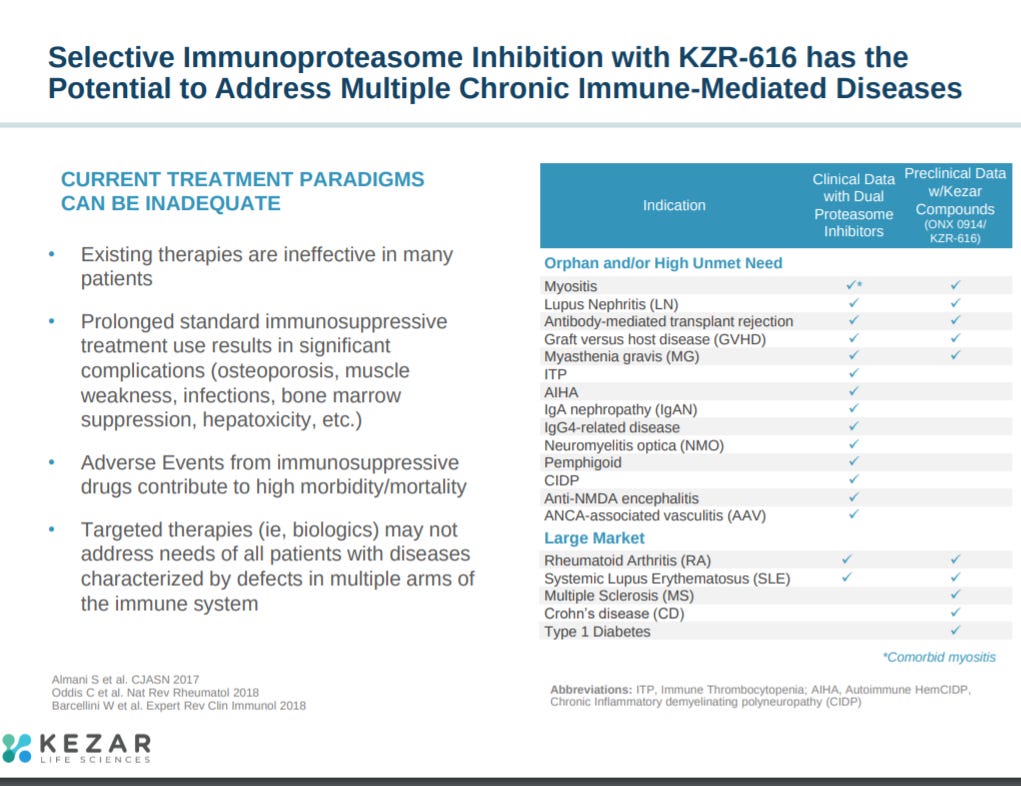
After this, the company explains their biological hypothesis. Inhibition of the immunoproteasome modifies the activity of the immune system without leading to cell death characterized by broader proteasome inhibitors.
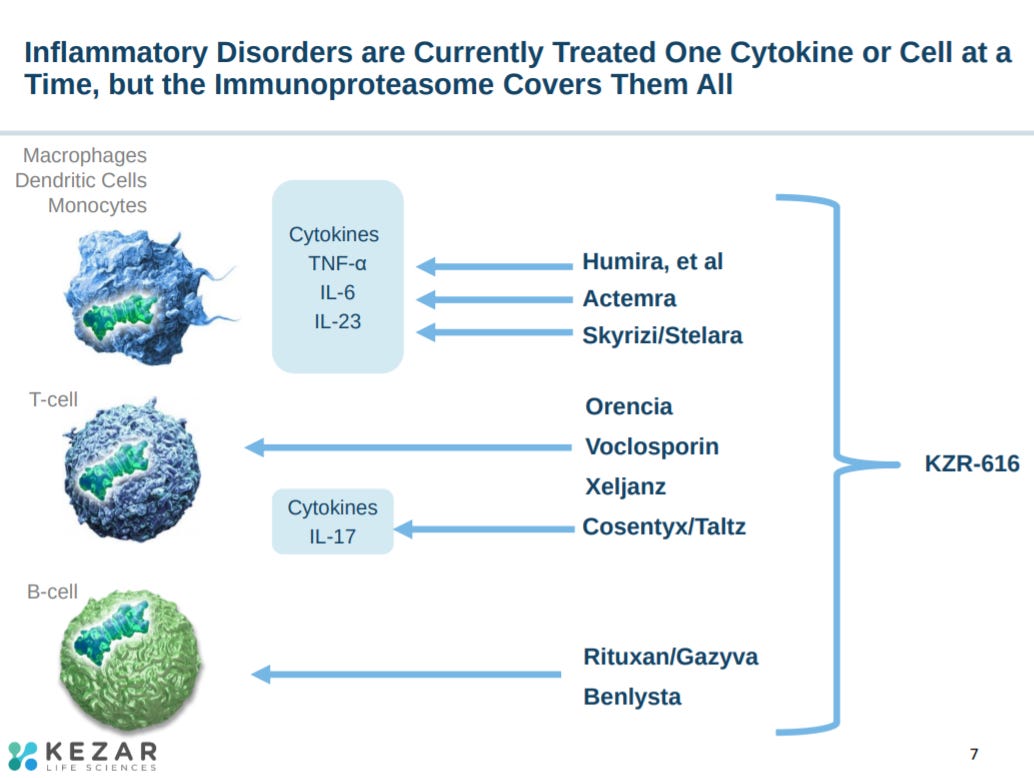
The immunoproteasome impacts a wide-set of immunology.
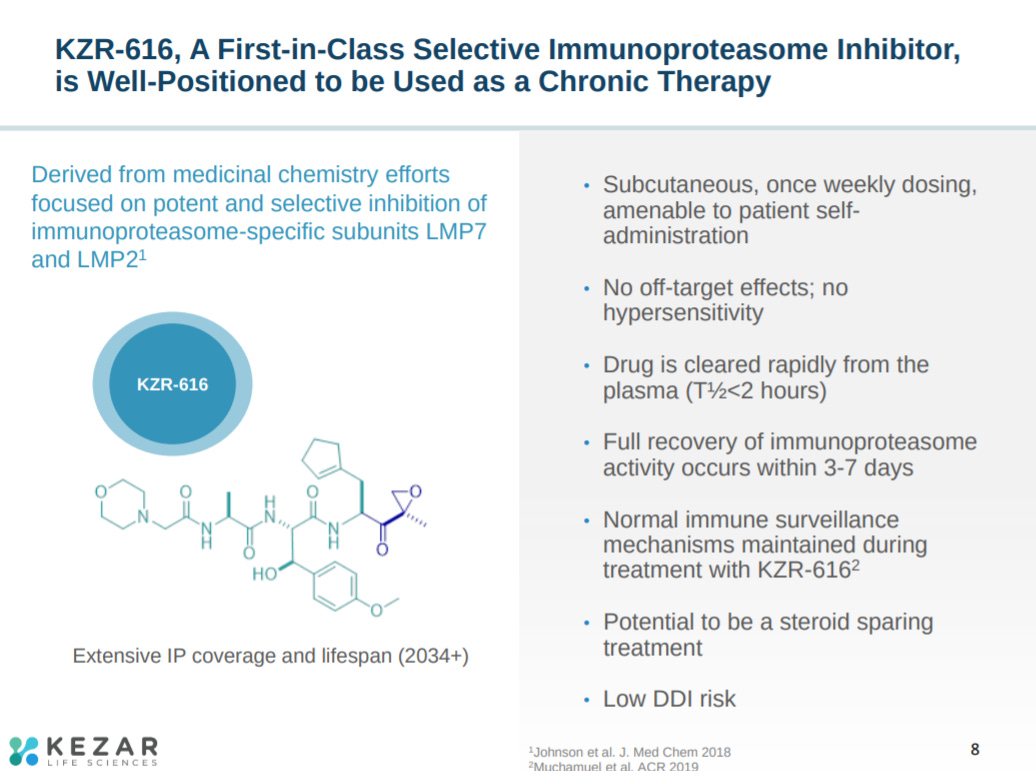
This slide explains Kezar’s lead asset, KZR-616. The molecule interacts with 2 subunits of the immunoproteasome.
With a MoA influencing almost every autoimmune indication.
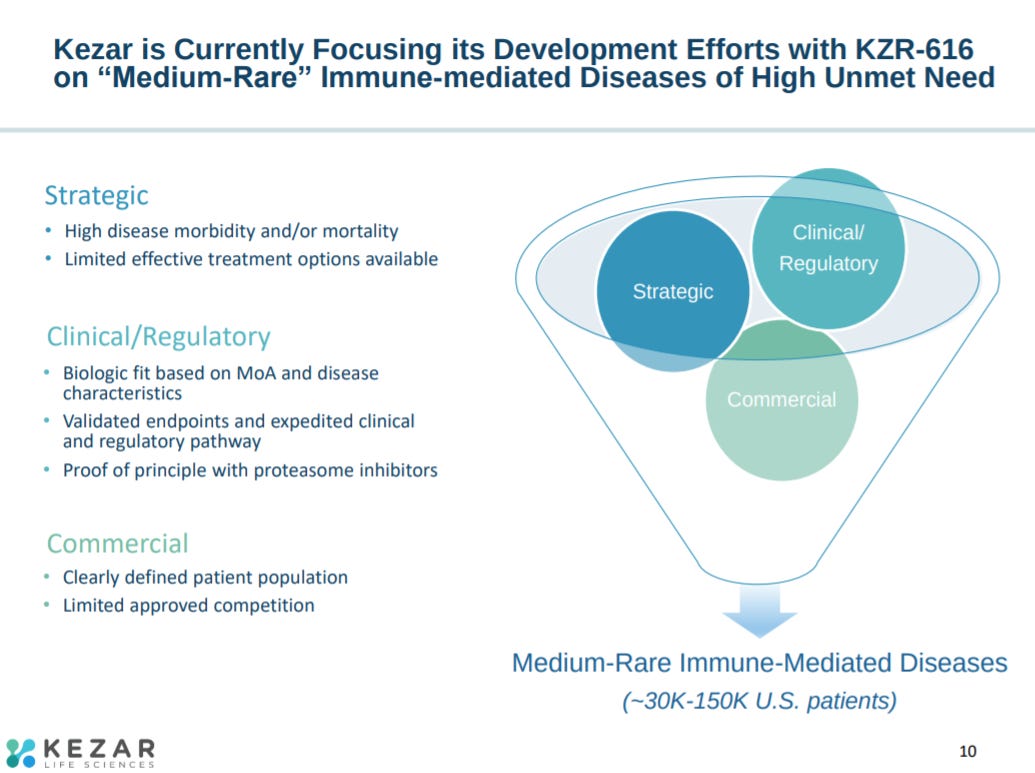
As a result, Kezar’s clinical strategy is rare-to-common. Pursuing diseases with smaller patient populations can accelerate clinical validation and set up KZR-616 to expand to larger indications like RA and MS.
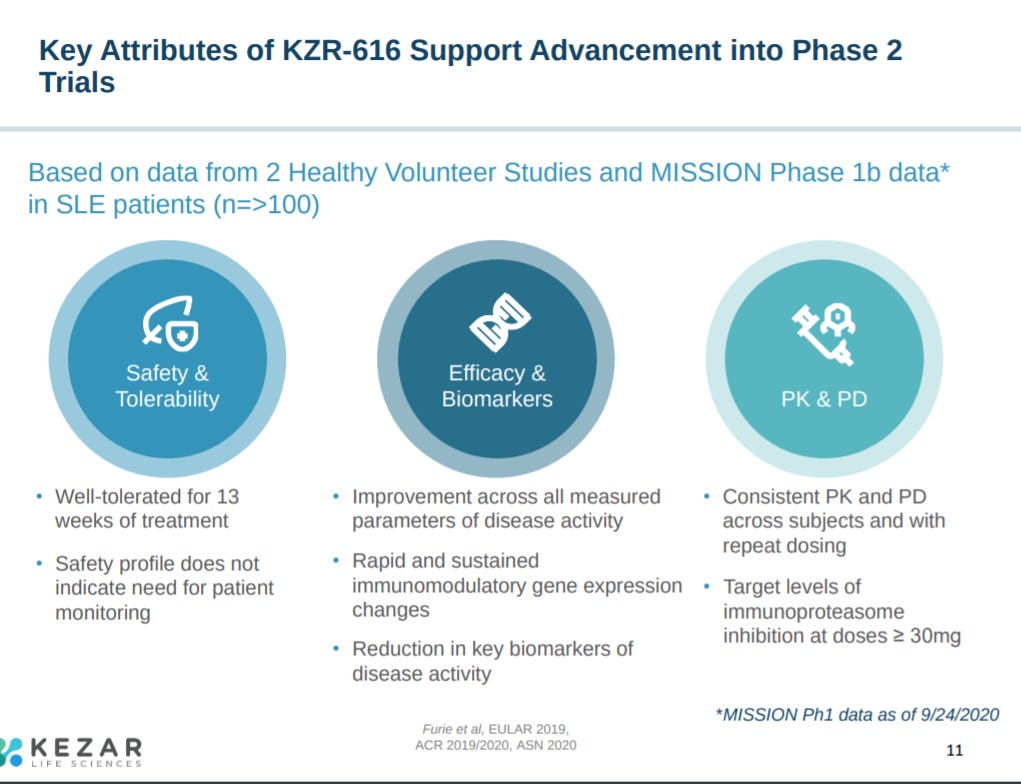
Current clinical data on KZR-616 showed safety up to 45 mg as well as immunoproteasome inhibition in humans.
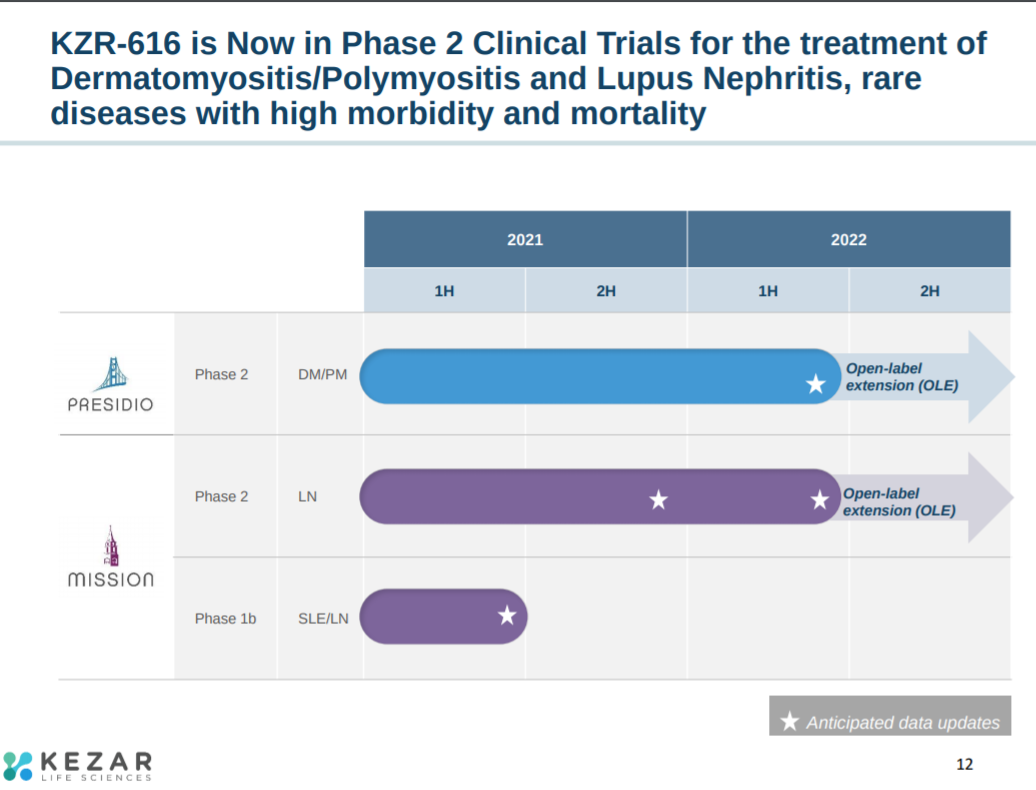
The clinical plan for KZR-616 is to initiate phase 2 trials in 2021 and expand down-the-line.
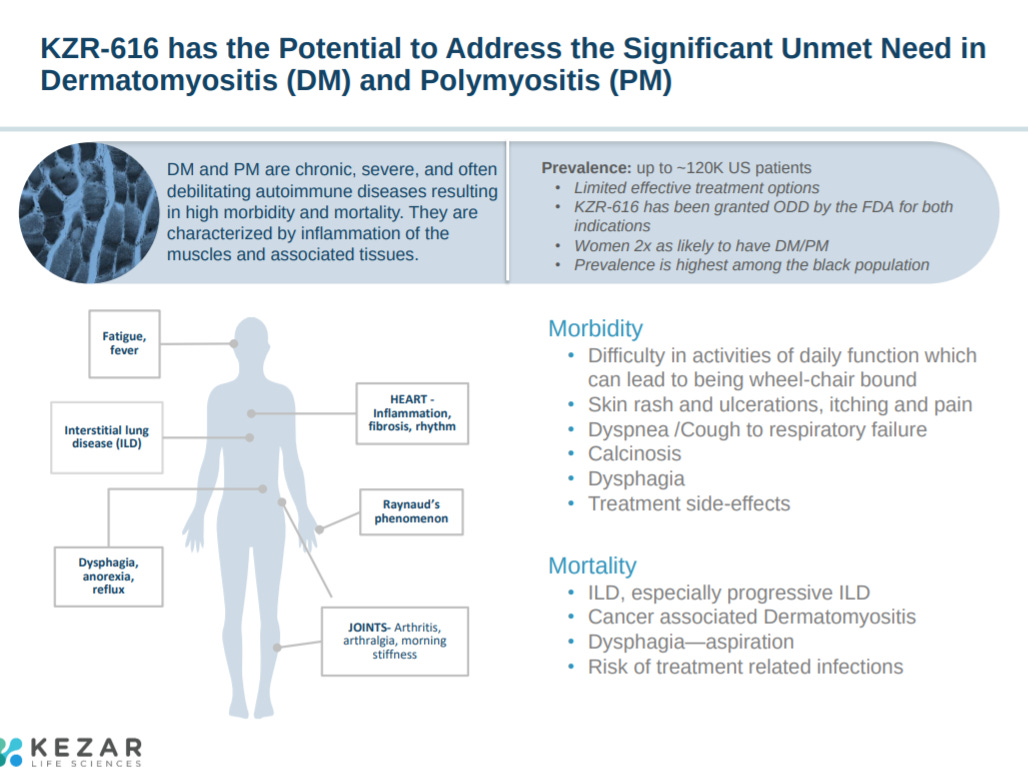
With an overview on 2 of the indications KZR-616 is focused on: dermatomyositis and polymyositis.
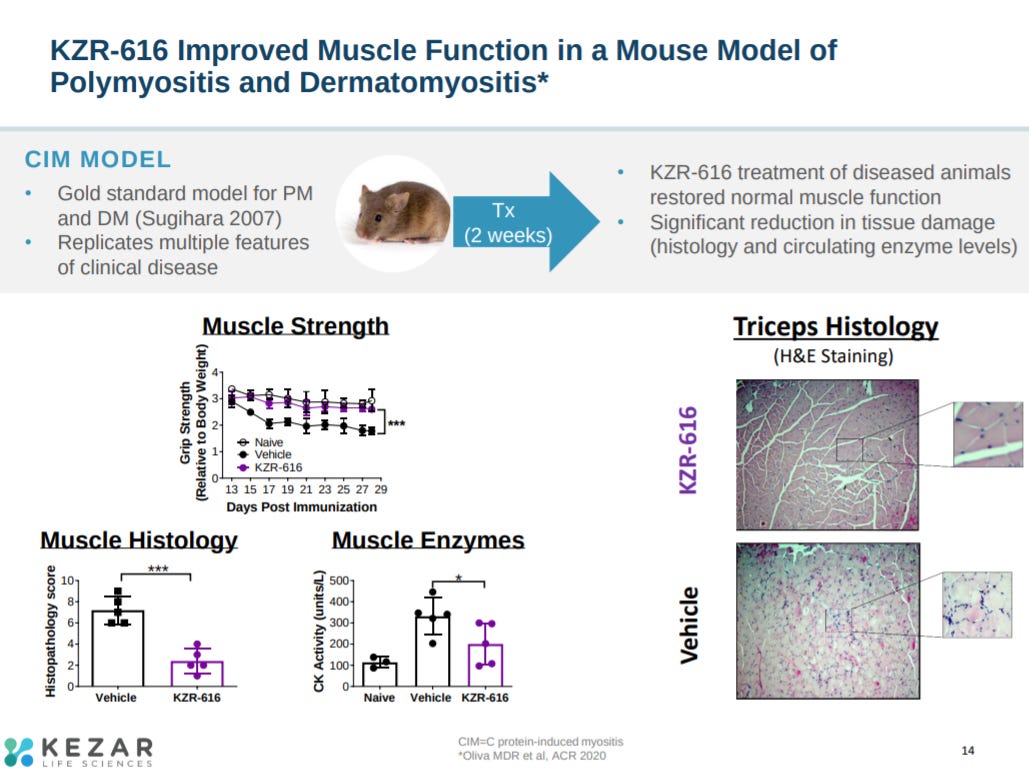
Data in a mouse model for dermatomyositis and polymyositis showed KZR-616 led to significant improvements in muscle function.
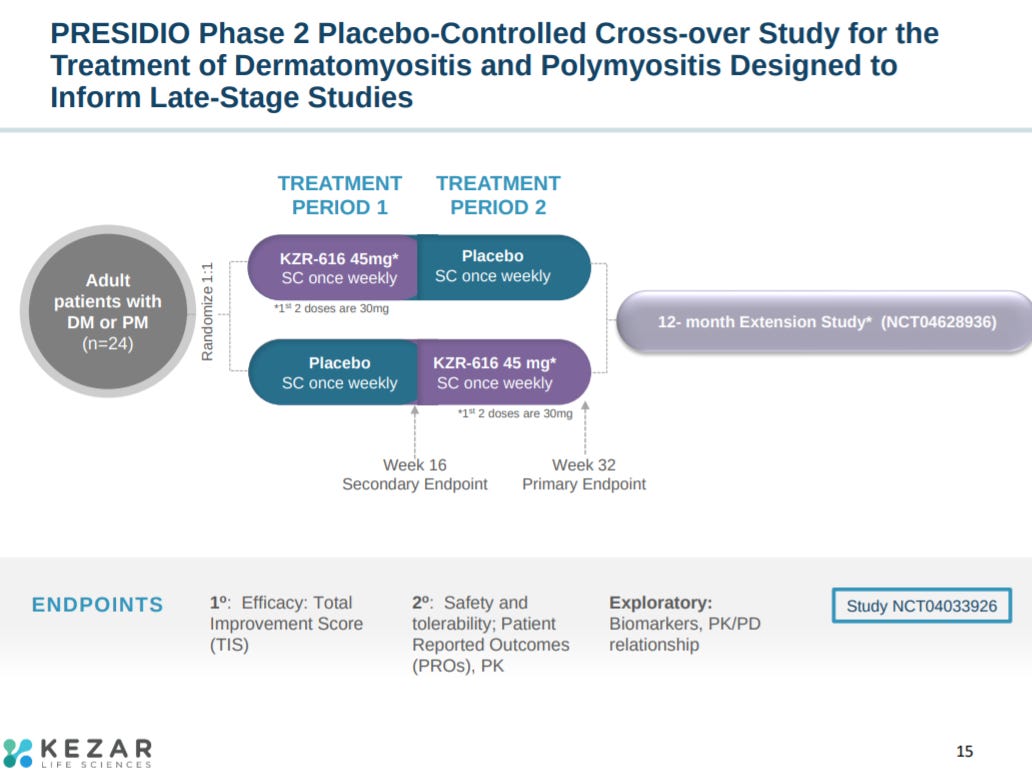
With an explanation of the phase 2 trial design for KZR-616 in dermatomyositis and polymyositis. Kezar established safety of the drug candidate in phase 1 trials for lupus patients.
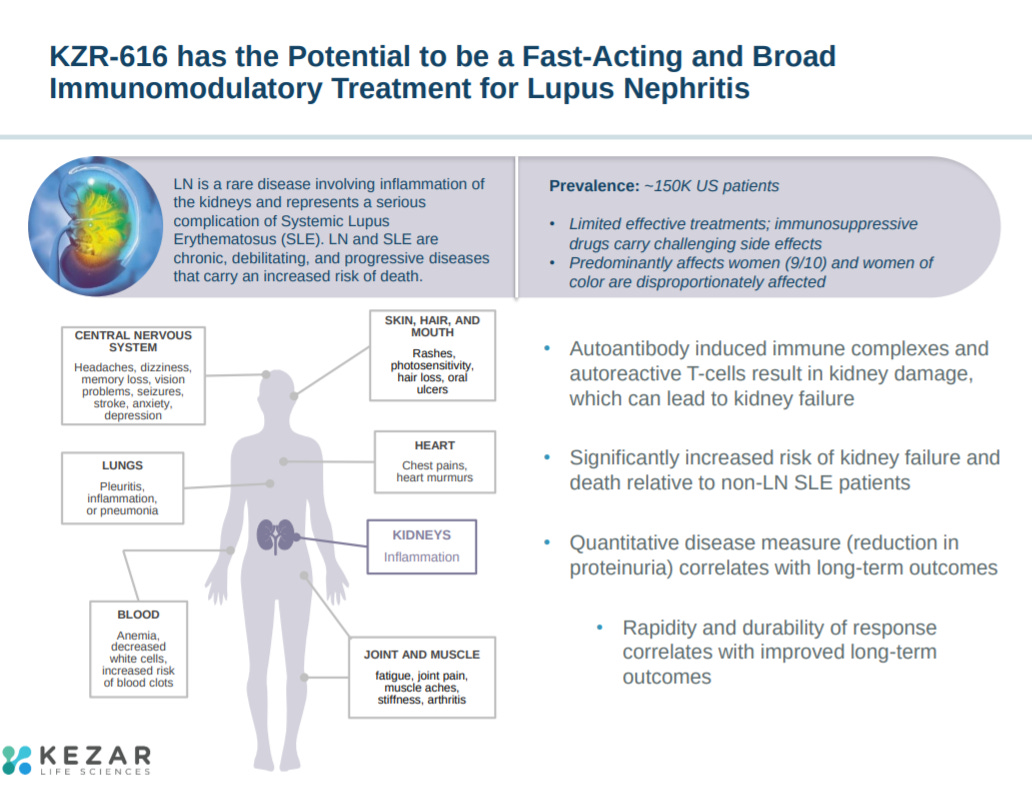
The company explains the potential opportunity in lupus nephritis (LN). Aurinia Pharmaceuticals just gained approval for voclosporin in LN - https://www.prnewswire.com/news-releases/lupus-foundation-of-america-congratulates-aurinia-pharmaceuticals-on-fda-approval-of-lupkynis-voclosporin-to-treat-lupus-nephritis-301213505.html
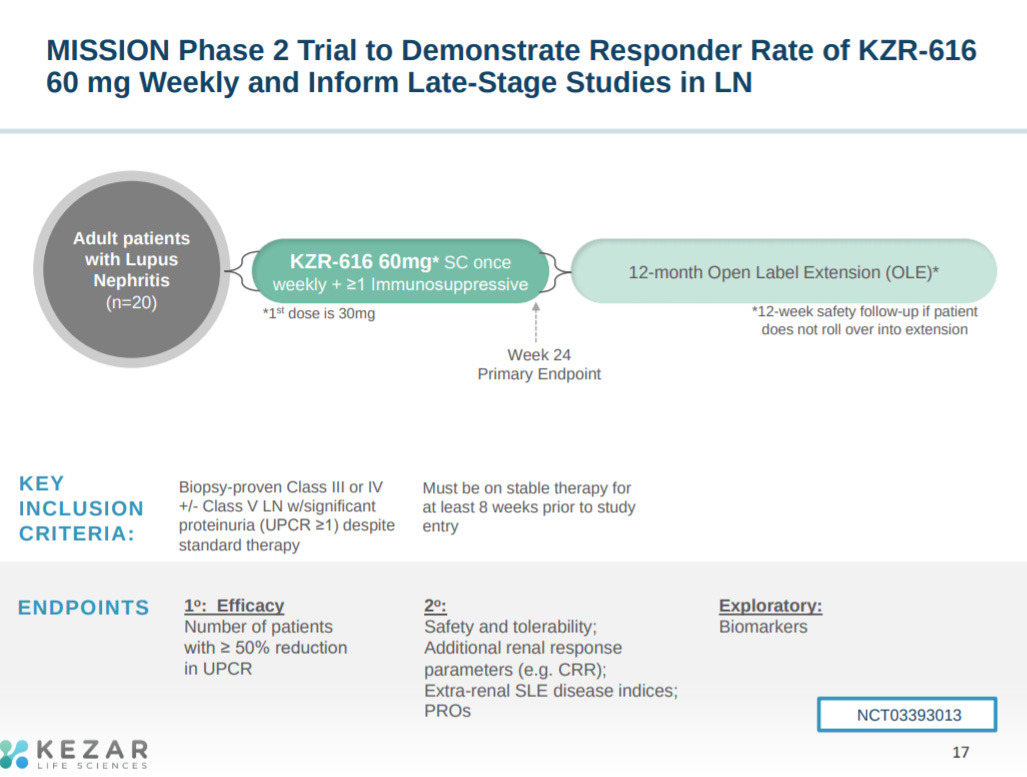
The next slide goes into the phase 2 trial design for KZR-616 in LN. They are going with the near the upper limit of dosing they’ve tested, which is a risk.
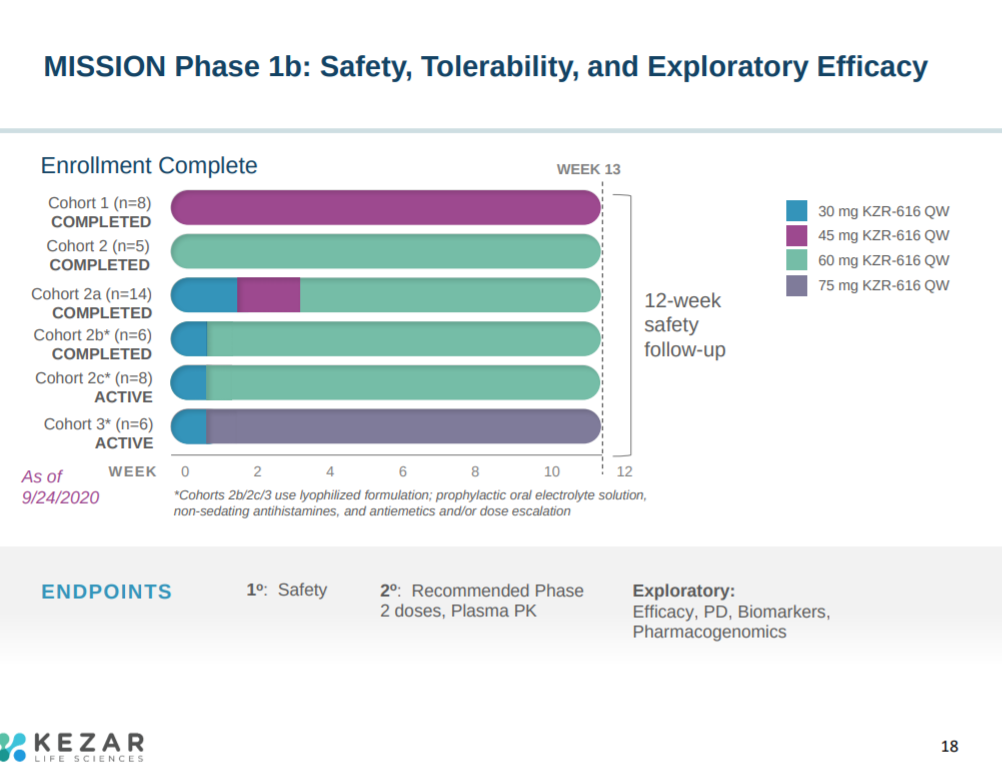
The phase 1 trial explored safety of KZR-616 at different doses: 30-75 mg.
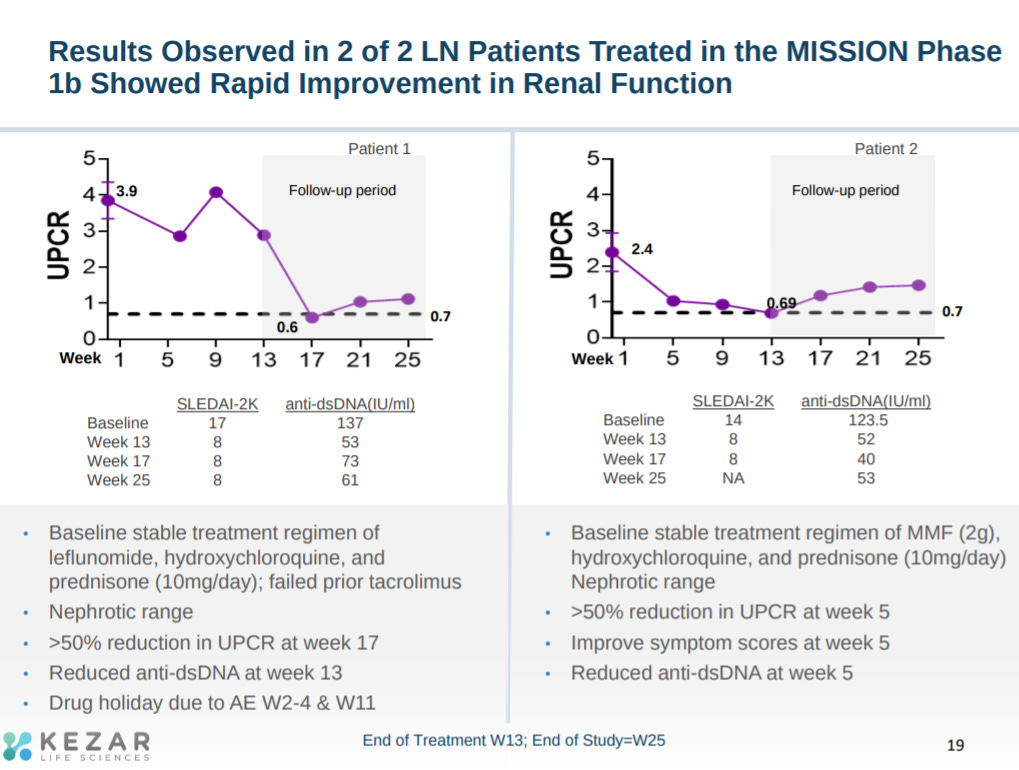
Phase 1 data, at least in 2 LN patients, showed pretty significant improvements in the phase 2 primary endpoint: urine protein/creatinine ratio (UPCR) reduction.
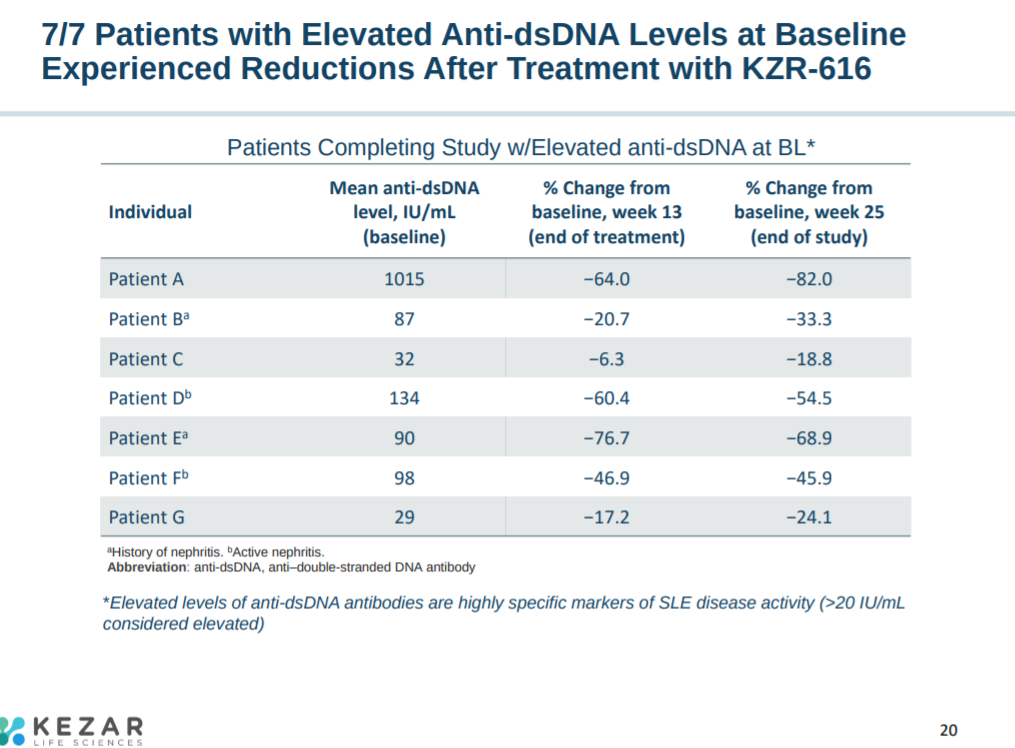
From the phase 1 trials, patients with elevated antibodies for dsDNA (a biomarker for lupus) all experienced significant reductions after KZR-616 treatment.
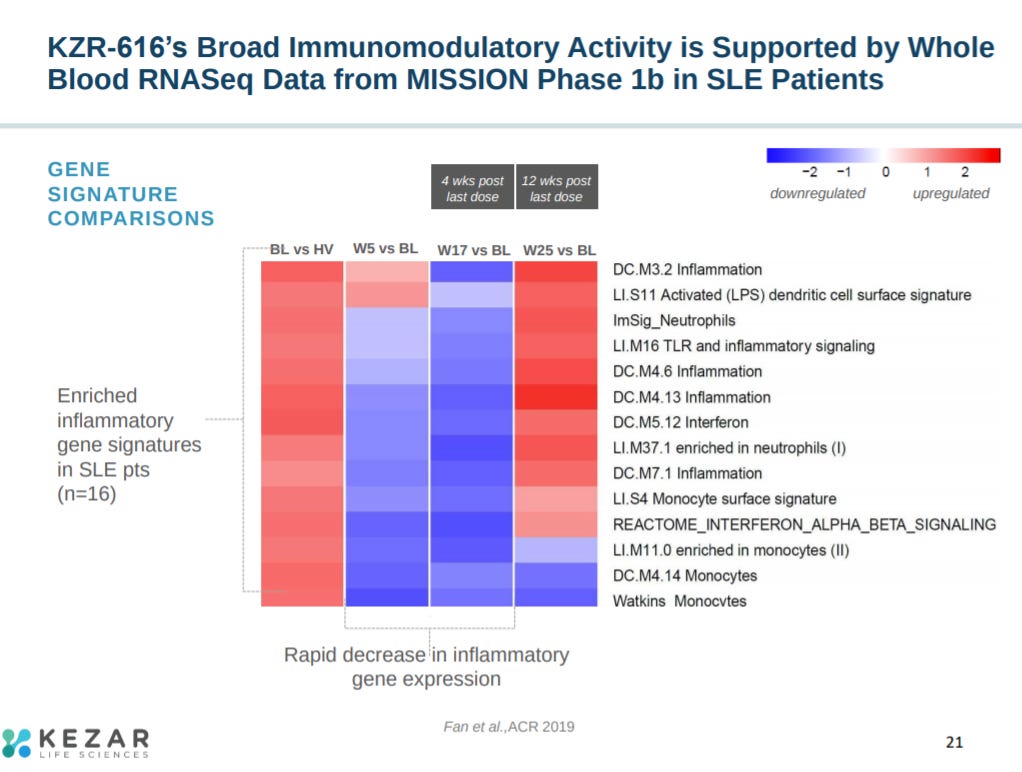
Phase 1 data showed a reduction in biomarkers involved in lupus 4 weeks after dosing KZR-616.
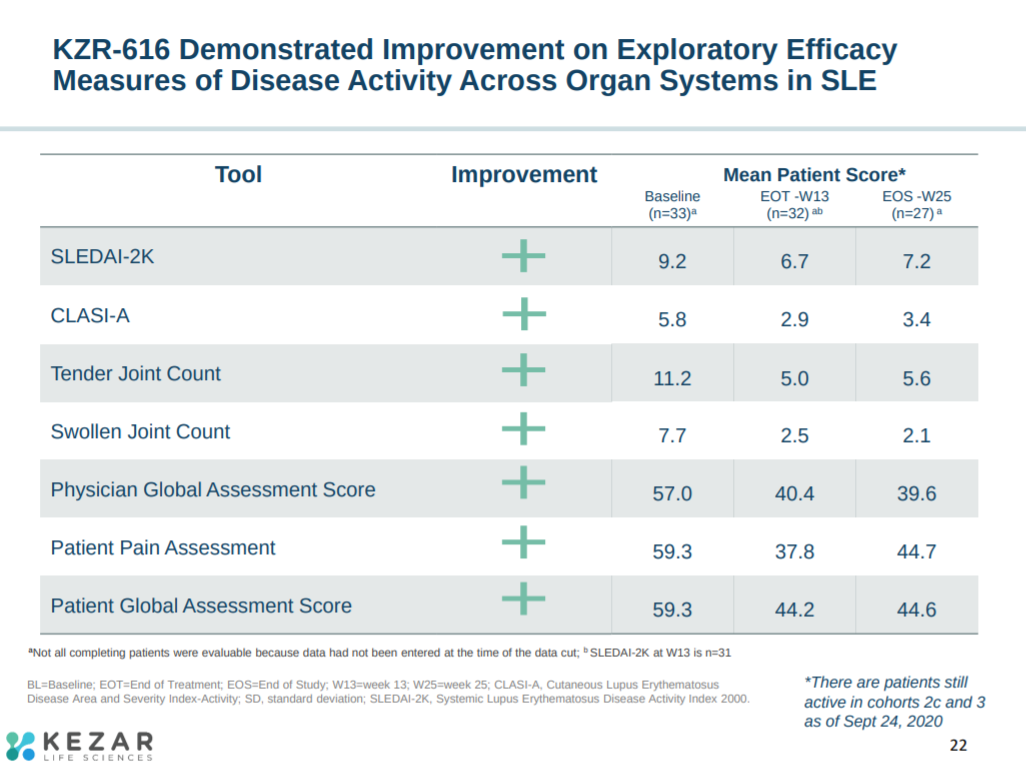
With improvements in secondary endpoints around pain and joint features (i.e. swelling).
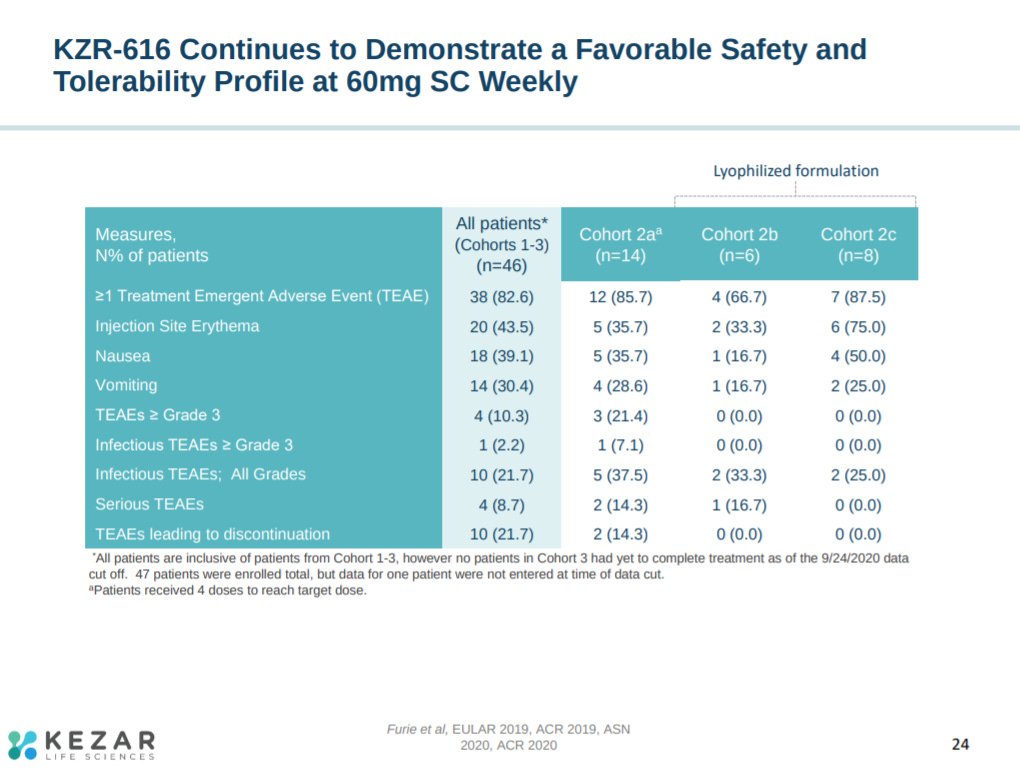
With a strong safety profile, but with 2 adverse events at 60 mg.
This slide provides a pretty useful table on KZR-616’s safety profile in the phase 1 trial.
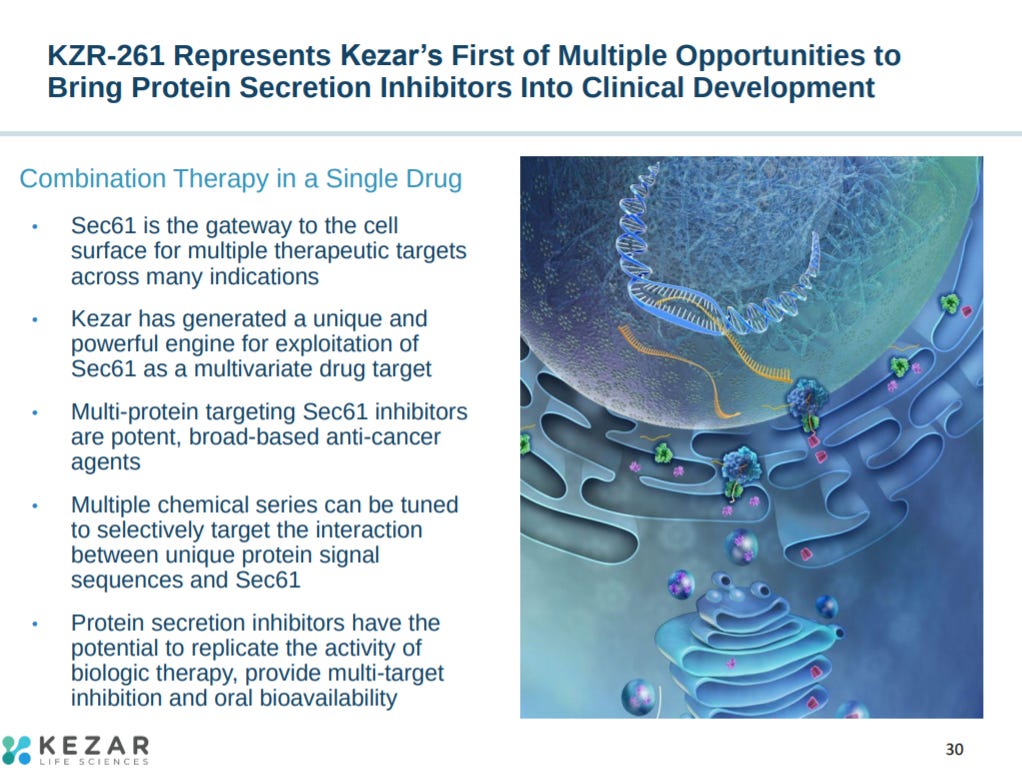
Kezar’s other asset targets protein secretion to treat solid tumors.
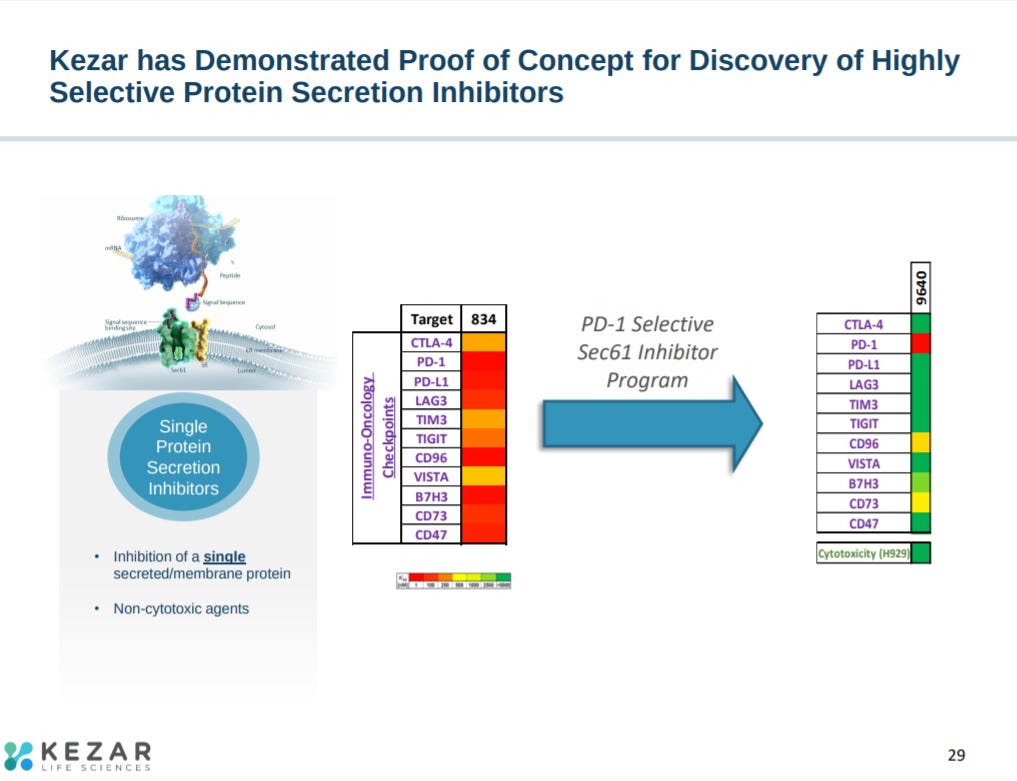
The biological hypothesis for targeting Sec61 is to have a wide-range to inhibit several proteins to one.
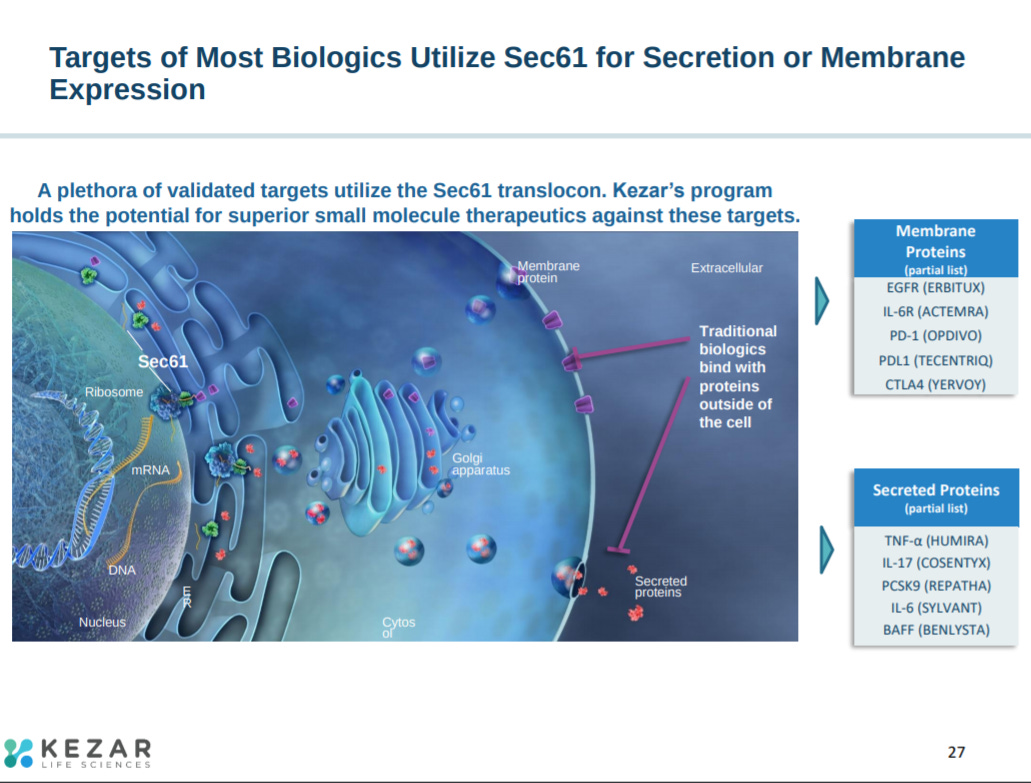
With many cancer targets relying on Sec61 to properly function. Would be nice to expand on the potential off-target effects.
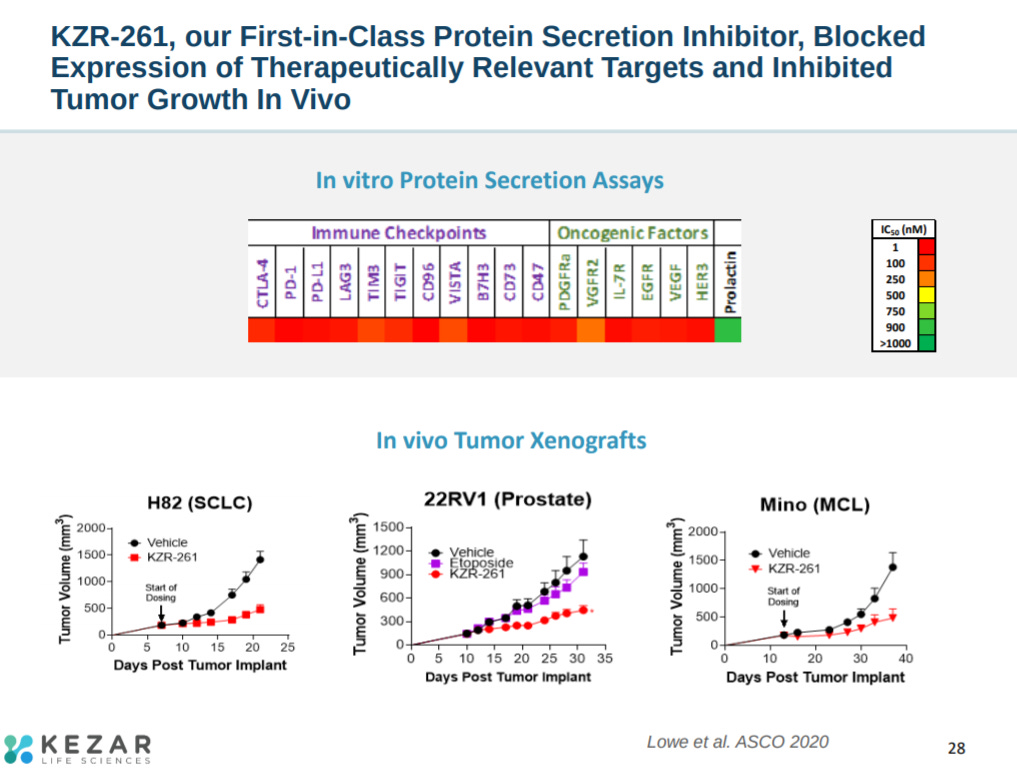
Preclinical data, in vitro and in vivo, showed lower secretion of important cancer targets and reduction of tumor volume in certain mouse models.
With this data, Kezar is focused on Sec61 inhibition to reduce secretion of immuno-oncology (IO) targets.
This enables KZR-261 to have combination potential with other IO drugs.
The presentation ends with an overview on Kezar.
Kezar’s presentation does a good job to convey the pipeline-in-a-pill strategy for KZR-616 and the overall opportunity in autoimmunity. Their rare-to-common strategy is the standard business model here. It would be great to compare their drug candidate with the current standard-of-care (SoC) as well as going deeper into some of the safety data. Especially since their phase 1 data release drove the stock price down above 50%.
Follow up questions for the team:
What licensing or partnership opportunities exist for KZR-261? Pairing the drug candidate with Keytruda or Opdivo?
Who are your competitors in drugging the immunoproteasome? AbbVie and Principia Biopharma (now part of Sanofi)?
How does KZR-616 compare to Aurinia’s drug among others in lupus?