More well thought out work can be found at — https://axial.substack.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you — email us at info@axialvc.com
Provention Bio was founded in 2016 by Ashleigh Palmer and Francisco Leon who had previously founded Celimmune (acquired by Amgen). Picking up teplizumab (their lead asset now that is ~30 years in the making) an anti-CD3 antibody for Type 1 Diabetes (T1D) from MacroGenics was a major breakthrough for Provention. The company got Jeffrey Bluestone as a director and refocused the asset on at-risk patients rather than only early onset patients where MacroGenic previously failed in a phase 3 trial.
Phase 2 data (n=76) in NEJM showed that teplizumab delayed the onset of T1D in high-risk patients (family members of type 1 diabetics). Follow up studies confirmed this result: teplizumab extended the time to clinical diagnosis of T1D to ~5 years from around two years in the placebo group and reduced the risk of progressing to insulin-dependent T1D by a little over 50%.
The first slide of their latest corporate presentation conveys Provention’s focus on treating early-stage autoimmunity including T1D.
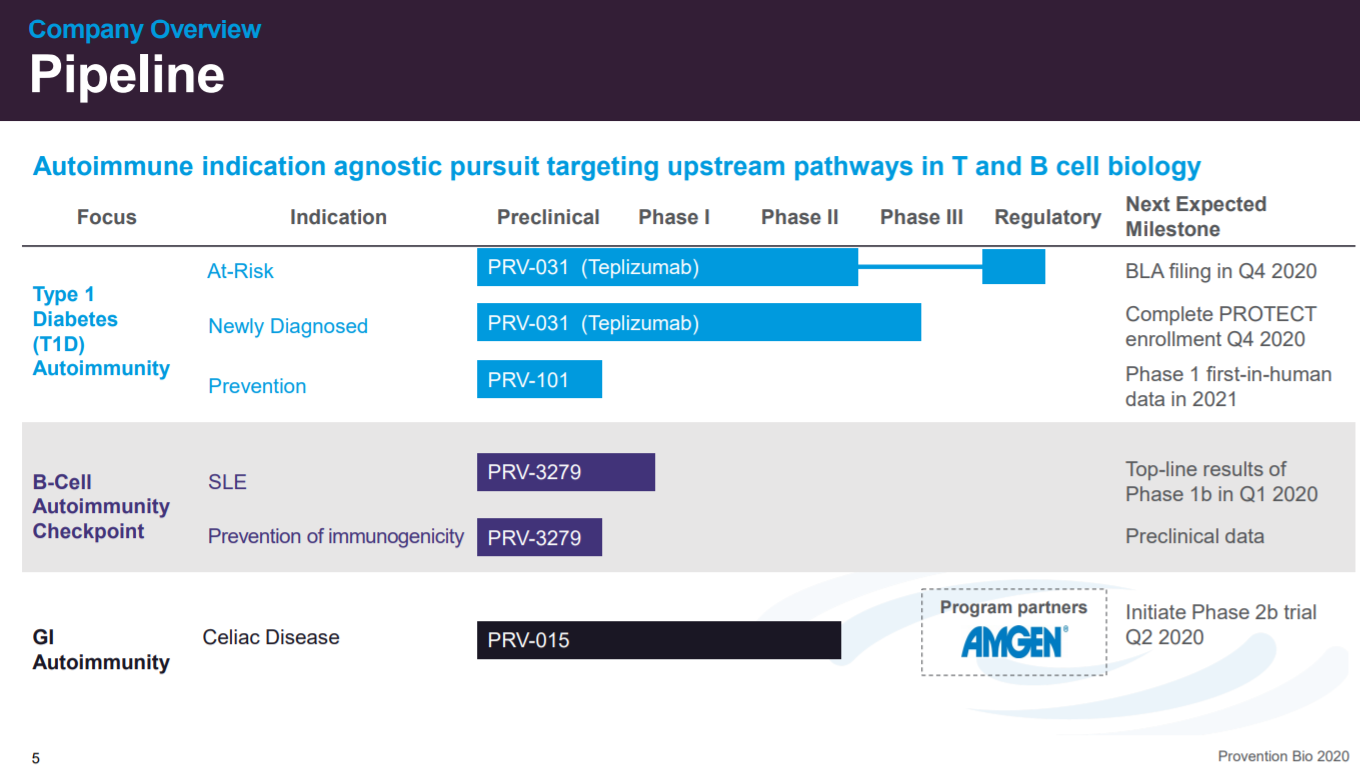
The next slide is an overview on Provention. Teplizumab (PRV-031) as the lead asset with other biologics and vaccines in development for T1D and other autoimmune conditions.
Provention is focused on early-stage intervention of autoimmunity.
The company's pipeline.
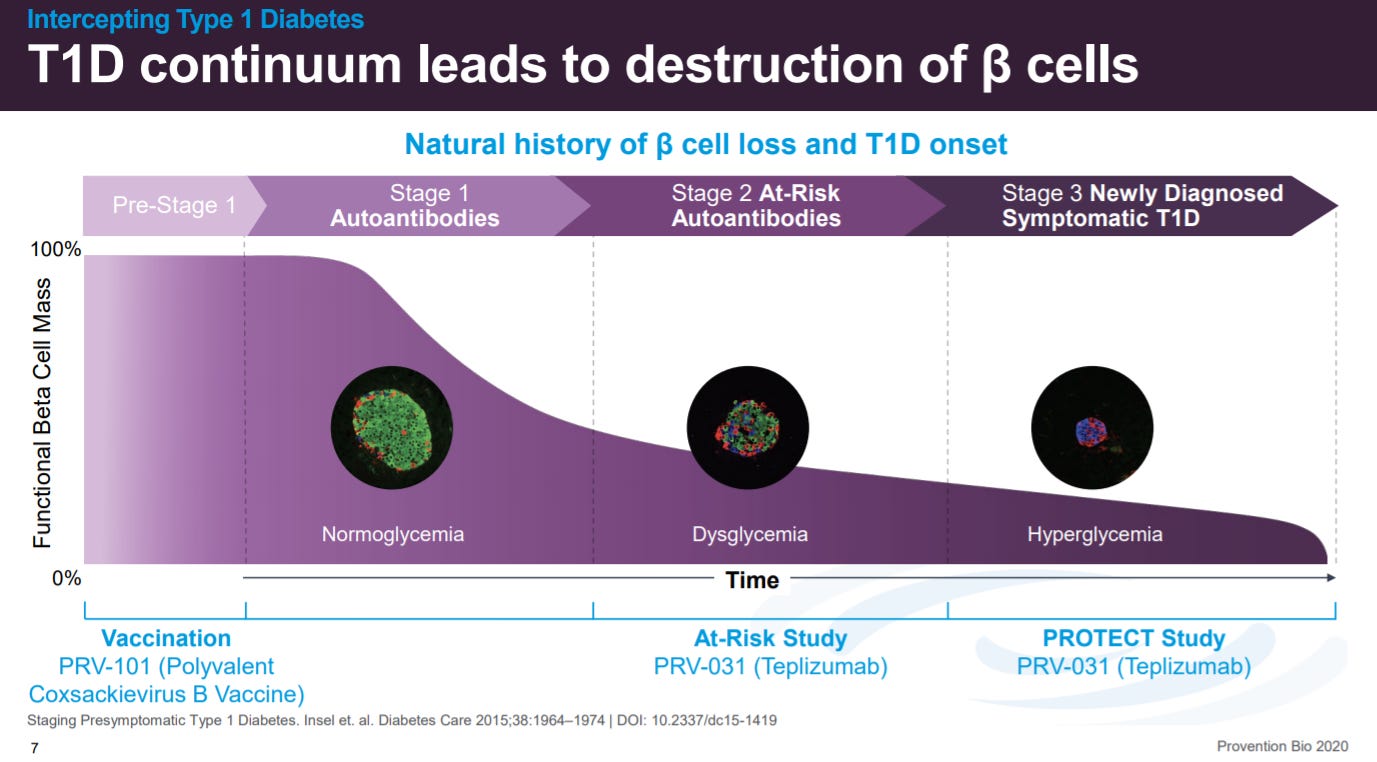
With an overview on T1D. Patients have not had a new medicine since insulin, almost a century ago, in the backdrop of incidence actually going up; maybe due to the hygiene hypothesis.
Across the stages of T1D, beta islet cell death is a consequence of the immune response, not the cause.
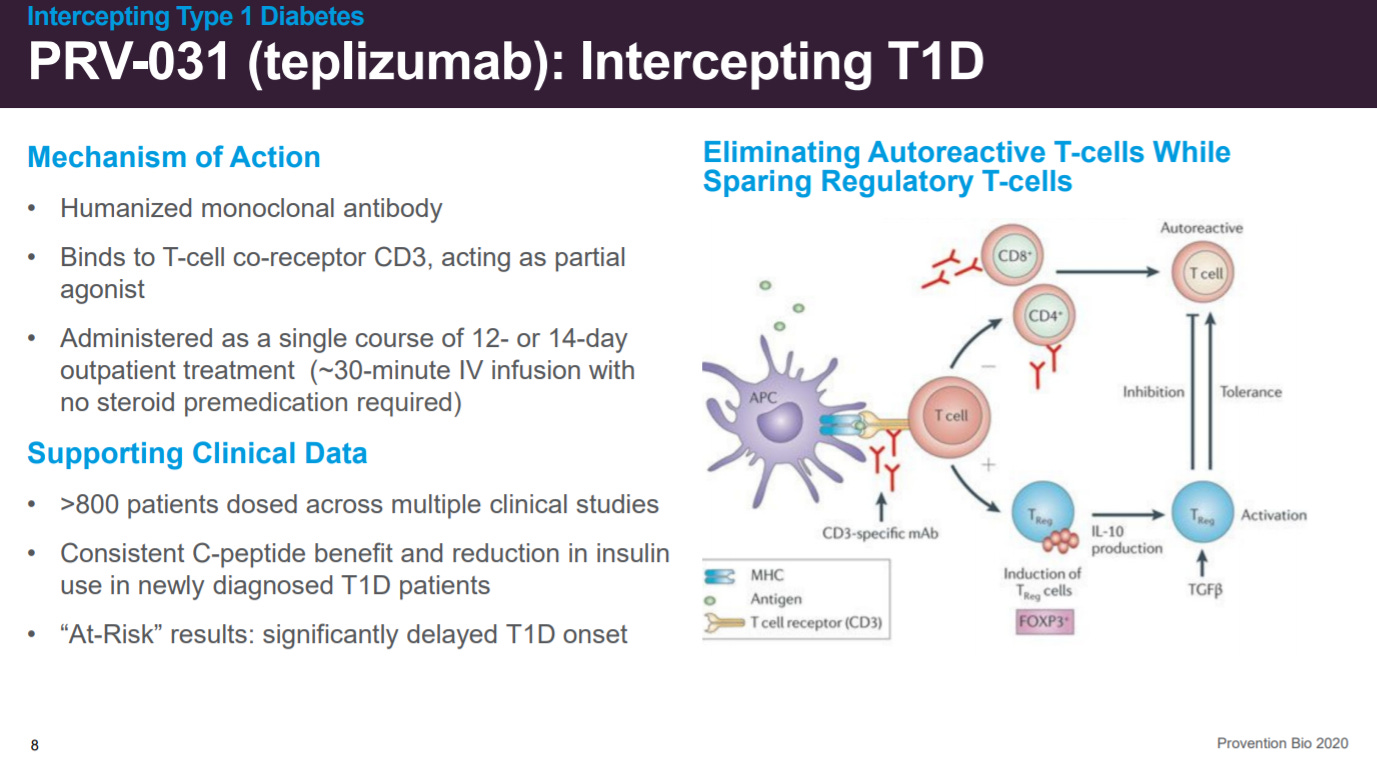
PRV-031 is a partial agonist to CD3 that has been dosed in hundreds of patients.
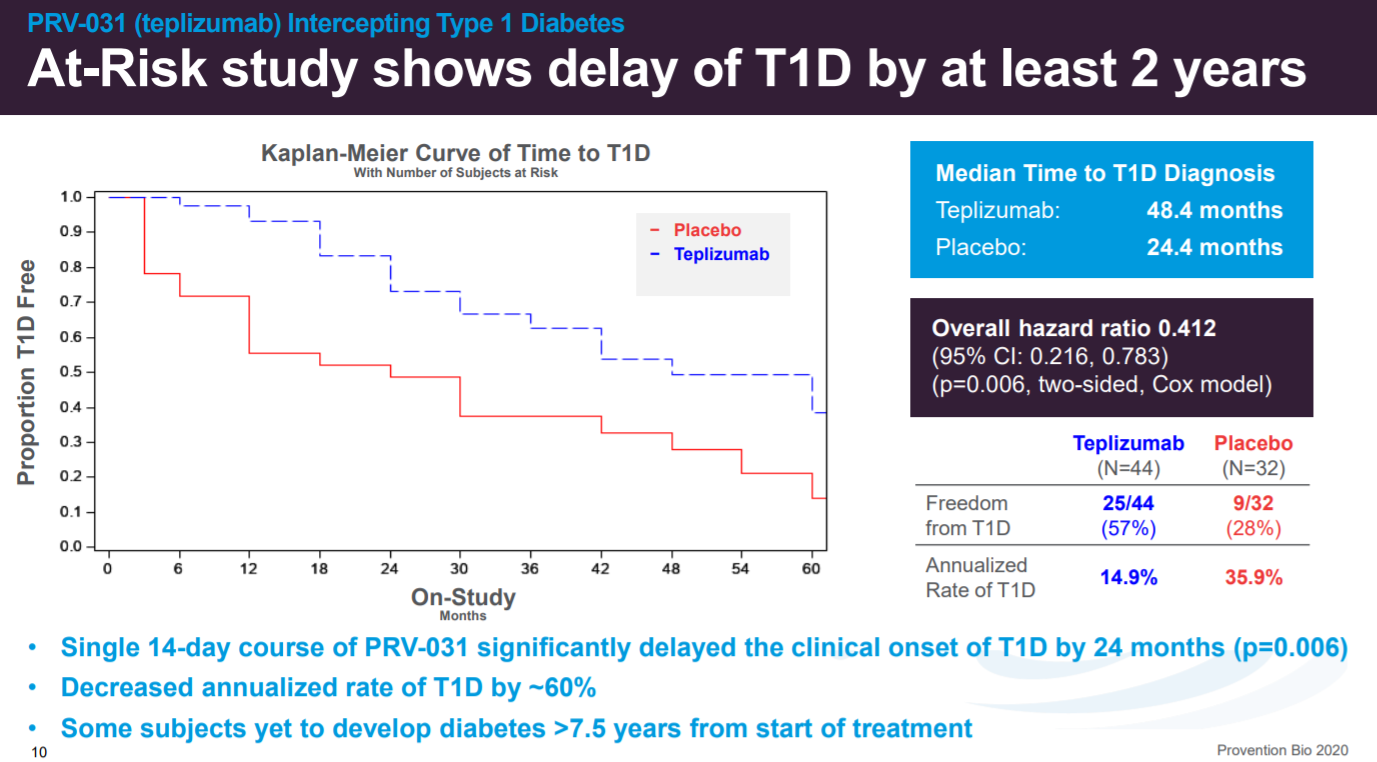
The important breakthrough for Provention and patients was the 2019 NEJM paper that showed that PRV-031 could delay the onset of T1D in at-risk patients.
In the clinic (n=76), extended the median time to T1D diagnosis by ~2x.
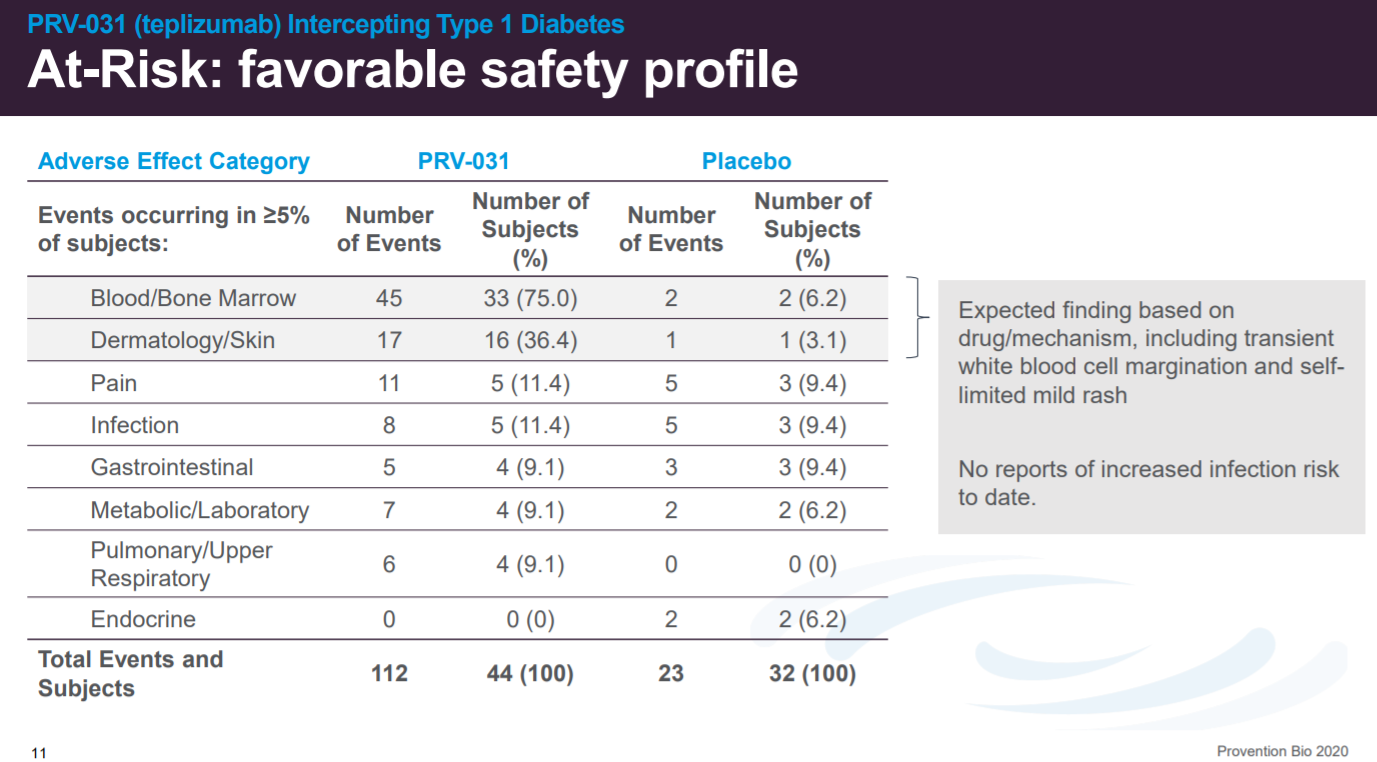
With adverse events associated with the bone marrow given that CD3 is involved in activating CD8+ and CD4+ cells.
Historical data on PRV-031 for early onset T1D patients has also shown some safety and efficacy.
For Provention’s lead asset, the company is working to get approval this year.
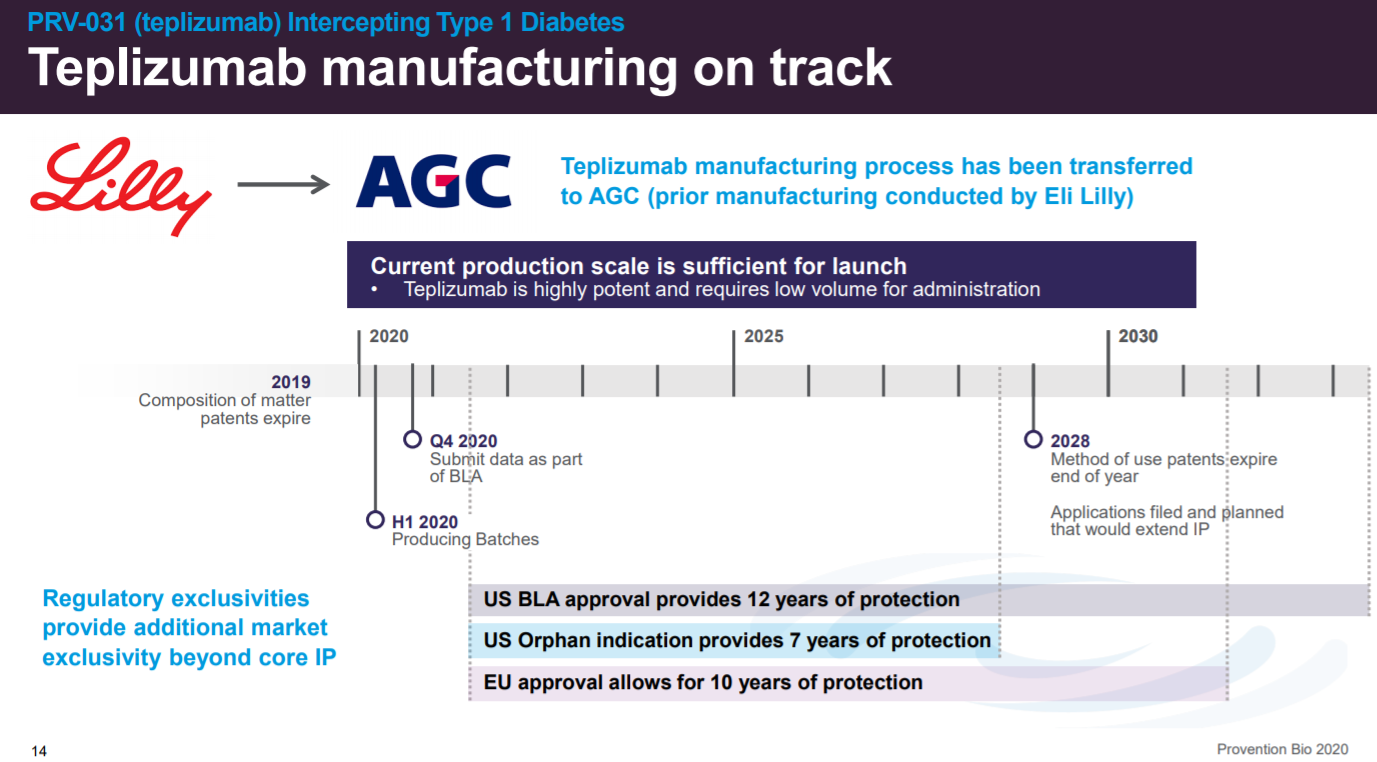
Where they are also lining up manufacturing with AGC, a CDMO.
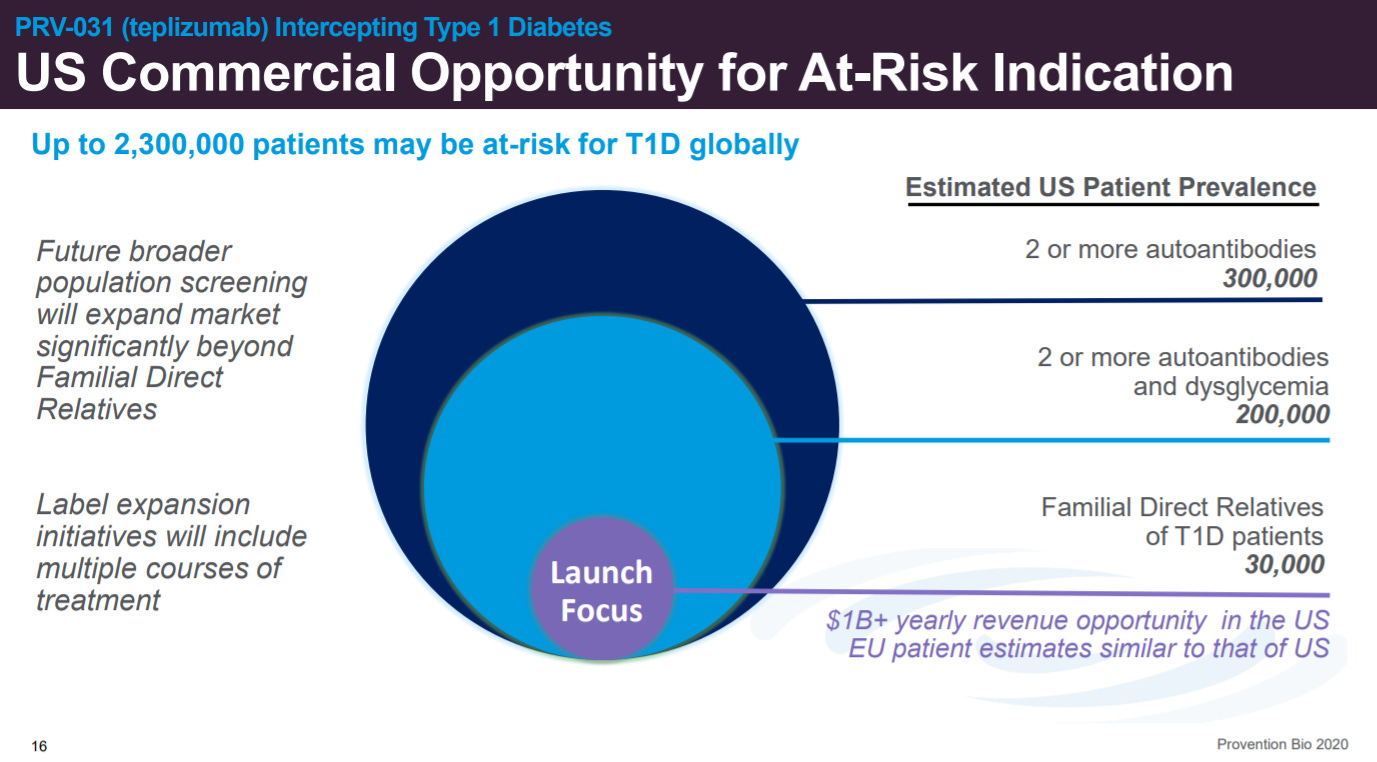
An important driver for Provention’s success will be patient screening and finding at-risk patients, not just those with early symptoms.
Provention will focus on patients with direct relatives that have T1D (~30K in the US)and expand to more patients as the screening gets better.
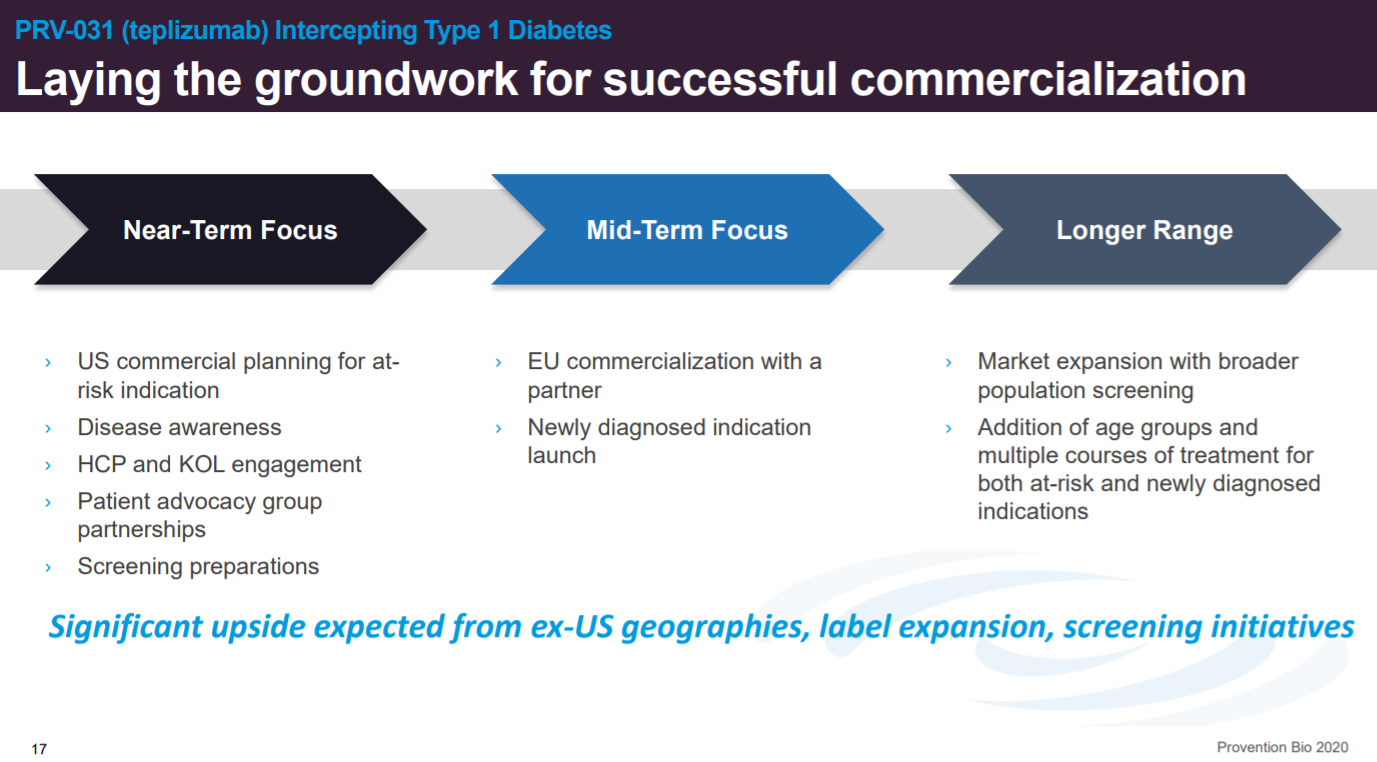
The company is focused on moving toward approval and setting up screening. Over the long-term, Provention wants to expand the patient population eligible for PRV-031 treatment.
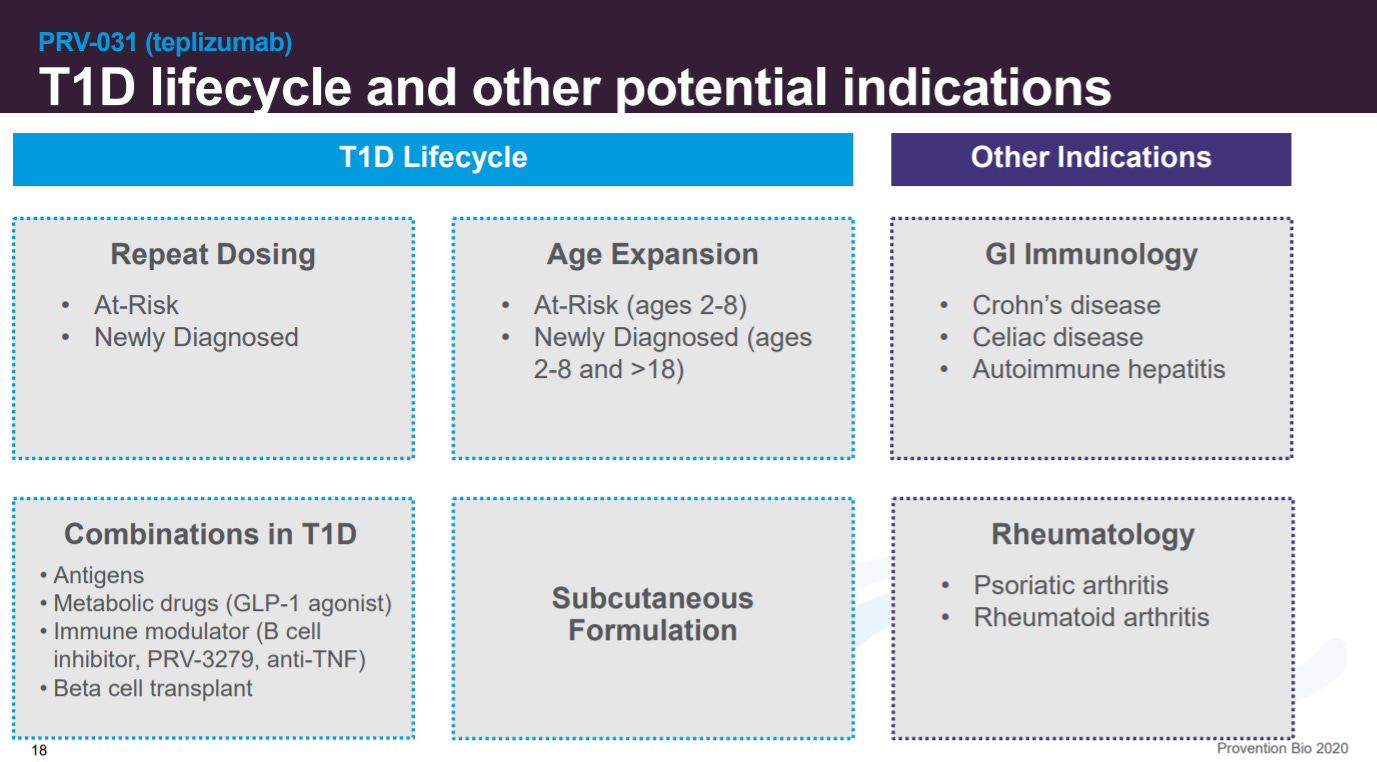
This slide goes into different strategies to expand PRV-031’s use in T1D and beyond.
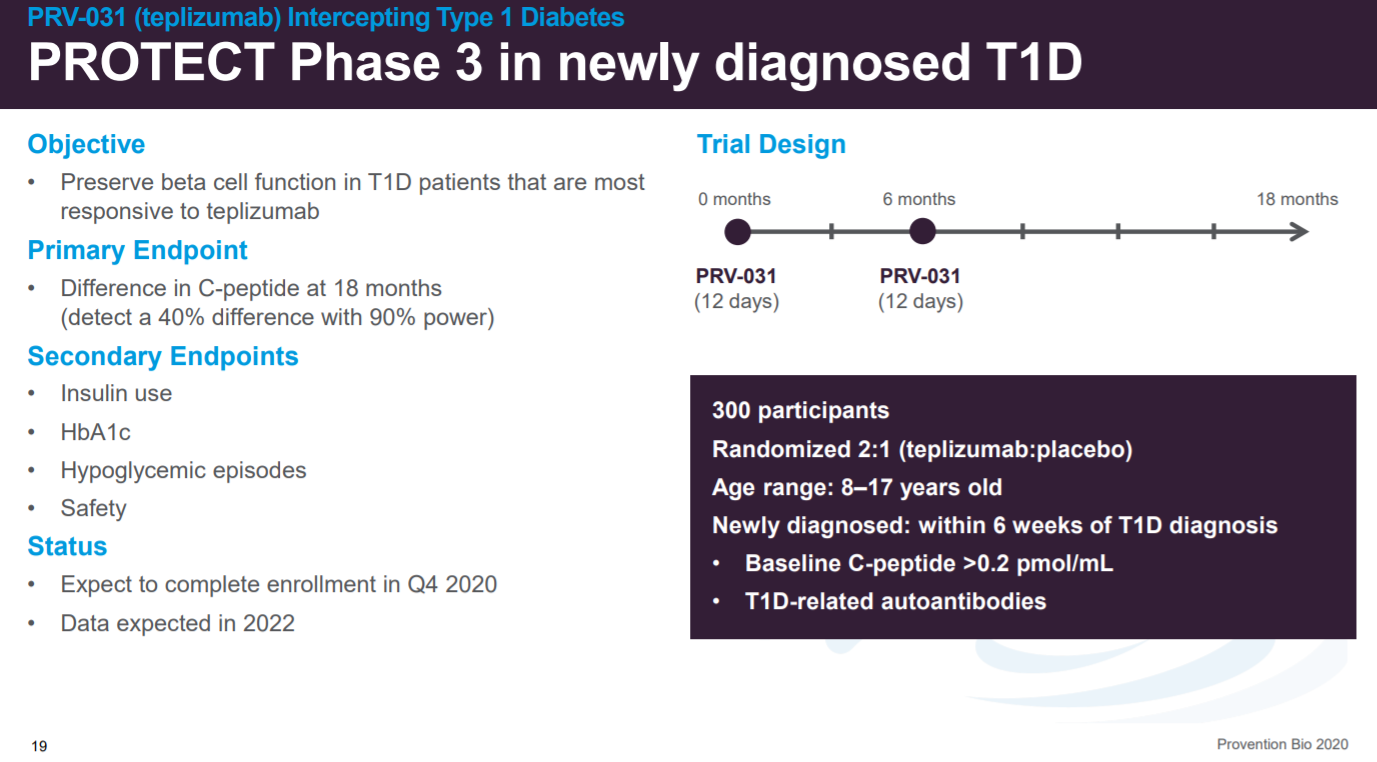
The phase 3 clinical trial (n=300) for the company’s lead asset in newly diagnosed T1D patients is to be completed in 2022.
With newly diagnosed T1D patients representing 40K new patients per year in the US.
Provention also is working on a vaccine (inactivated CVB) for T1D and celiac disease.
With phase 1 clinical trials initiated and data expected later this year.
The company has a bispecific program to inhibit the production of autoantibodies in autoimmunity.
This program is undergoing a phase 1/2 clinical trial with the trial expected to start in lupus in 2H2021.
Provention also has an anti-IL15 antibody for celiac disease patients.
A slide on PRV-015’s phase 2 clinical trial design.
As of last year, Prevention had enough cash (given that it is still virtual) for 1-2 years and get to pivotal trials for its lead asset.
A team slide. The founders still run Provention.
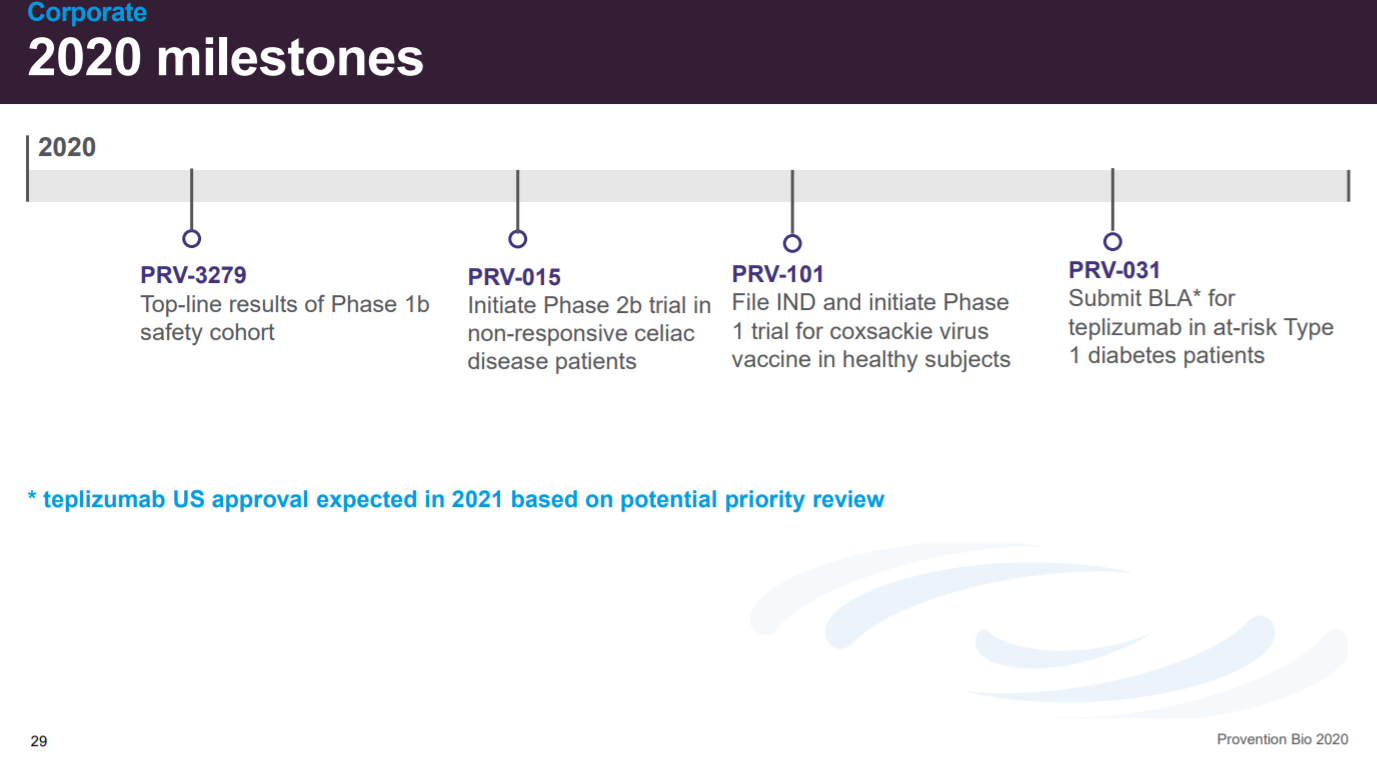
The penultimate slide is focused on milestones with a focus on PRV-031.
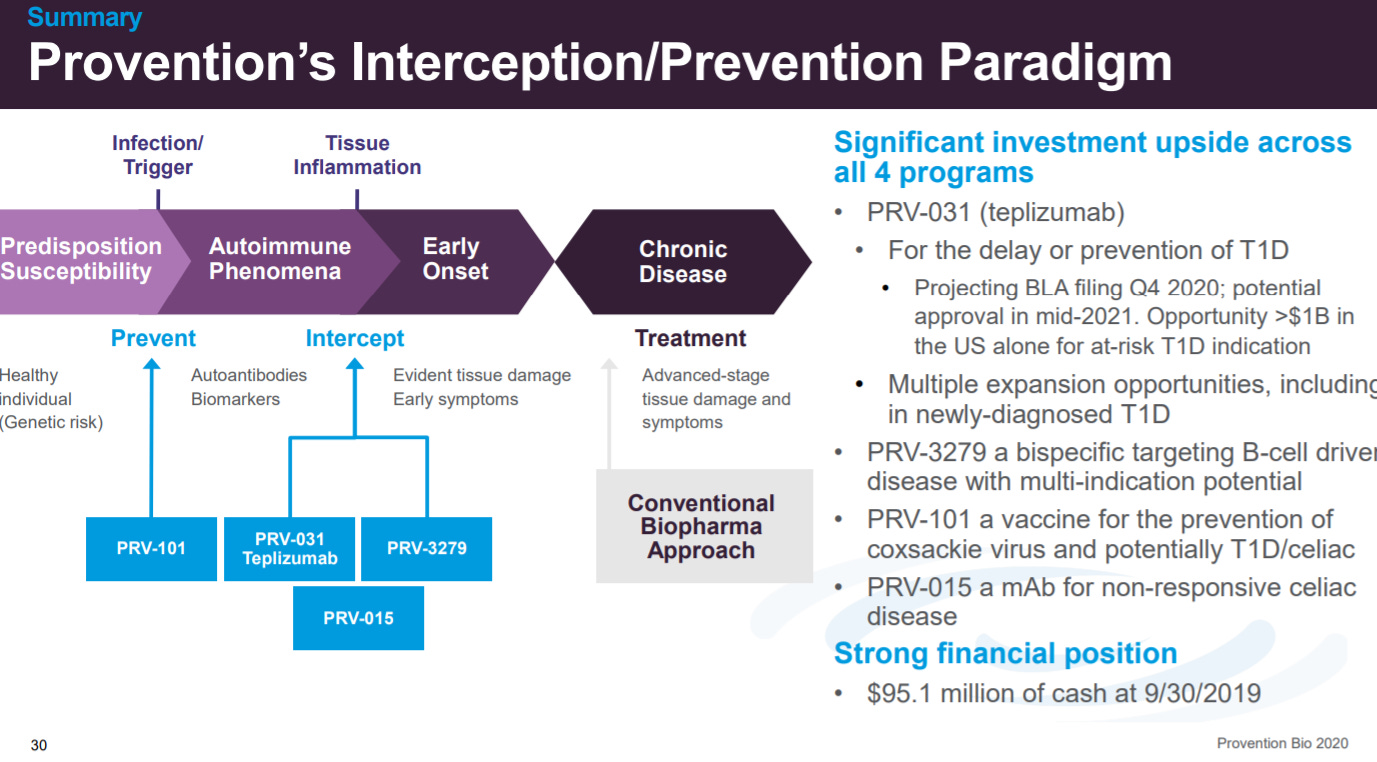
The last slide positions Provention’s drug candidates on the earliest stages of autoimmunity.
Provention’s presentation does a good job to frame their focus on early-stage intervention. For T1D, there is a massive patient need for new treatments, and Provention’s data on PRV-031 ability to delay disease onset in at-risk patients could be transformational. The key issue here is patient screening and finding which patients would benefit the most.
Follow up questions for the team:
How is Provention working toward scaling up patient screening? Partnership with a company like Invitae?
For the ~30K at risk patients in the US, how does reimbursement work? Standard and similar to patients with T1D?
How long will the label expansion take for PRV-031?