More well thought out work can be found at — https://axial.substack.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you — email us at info@axialvc.com
Sio Gene Therapies (formerly Axovant) was founded in 2014 as a Roivant company to develop gene therapies for neurodegenerative diseases. With a pipeline of drug candidates for Parkinson's disease, GM1 gangliosidosis, and Tay-Sachs disease and a recent name change and pivot, Sio is making exciting progress to bring gene therapies to the brain.
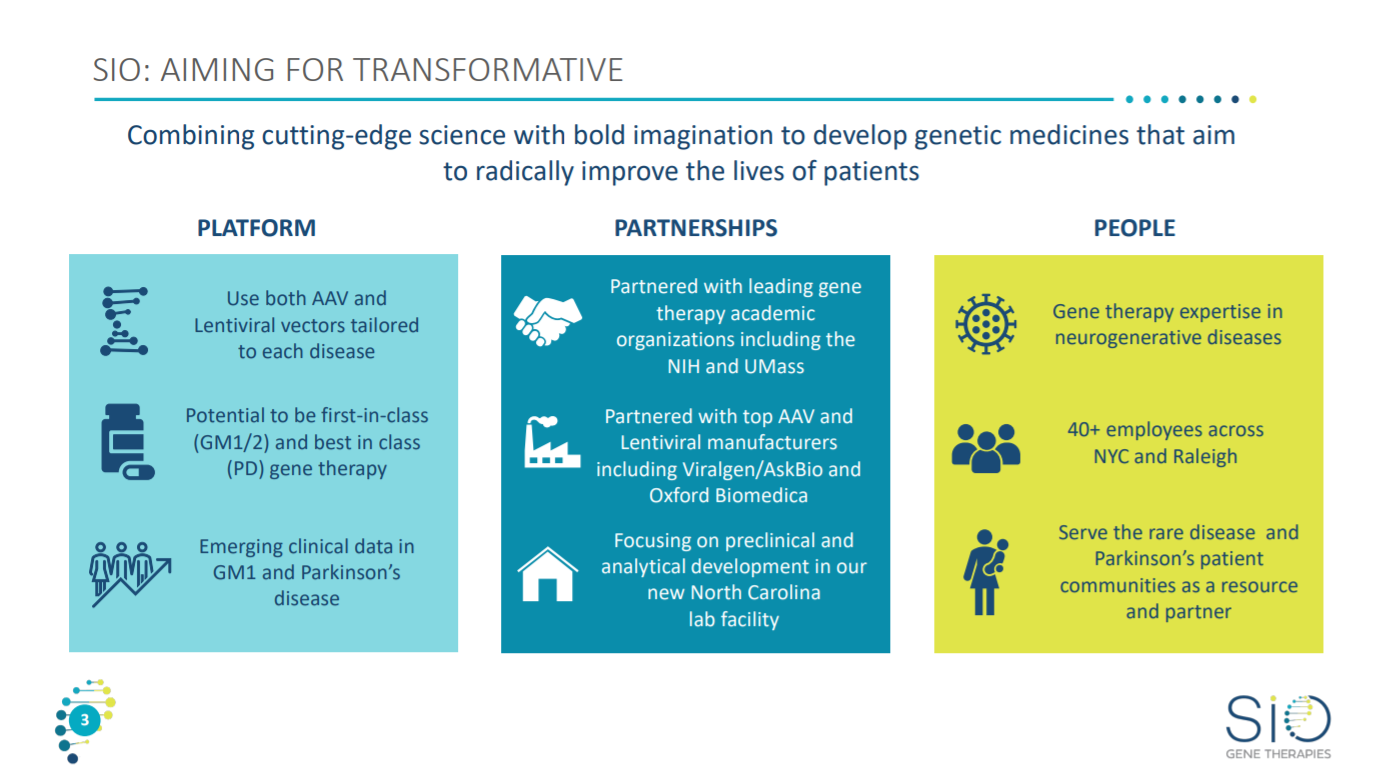
The first slide of their latest corporate presentation gives an overview on Sio’s platform and business model.
The next slide is on the company’s executive team.
Then onto their pipeline.
After introductory slides, the company goes into their lead program for GM-1/2 gangliosidosis.
In GM1 gangliosidosis, the disease is caused by a mutation in the GLB1 gene and Tay-Sachs is driven by mutations in the HEXA/B genes.
Natural history studies show high mortality rates for these patients a few years after birth.
As a result, Sio is working to develop gene therapies to replace GLB1 and HEXA/B.
The company’s lead gene therapy program is focused on GM1 gangliosidosis. There are around 400-900 patients in the US and Europe with no currently approved treatments.
The gene therapy is an AAV9 construct designed to restore functional copies of GLB1.
With the potential to make a large impact for patients in the late-stages of the disease.
Delivery through IV allows for Sio’s gene therapy to have broad distribution in the body since GM1 gangliosidosis affects multiple organs over time.
In GM1 cat models, Sio showed an ability to restore normal activity based on a clinical rating score - https://www.cell.com/molecular-therapy-family/molecular-therapy/pdfExtended/S1525-0016(17)30015-1
Showing broad distribution of the AAV gene therapy.
As well as showing a significant increase in survival for IV delivered AAV9.
This slide synthesizes this preclinical data package.
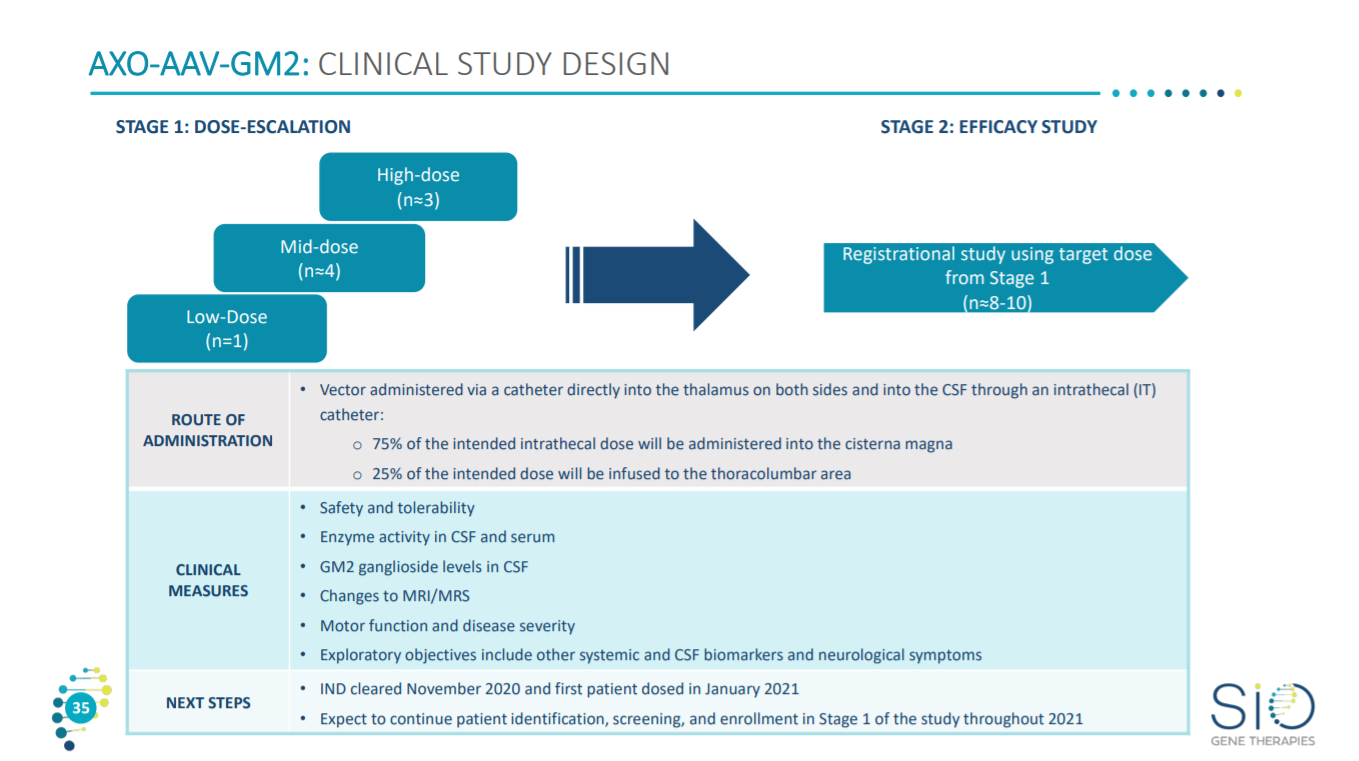
For the phase 1/2 clinical trial, Sio is working to determine the safe dosing range and get early efficacy signals in humans.
The endpoints for the trial are based on a clinical score and biomarkers for GLB1 activity.
The next section of the presentation goes into the initial clinical data for Sio’s lead program.
At the low dose, the gene therapy has been found safe so far in earlier stage patients (n=5).
Initial data shows a ~2x increase in beta-galactosidase activity from baseline after 6 months. Pretty exciting.
With an increase in enzyme activity to 20%-50% normal after 6 months.
This slide is a summary of the initial data.
At 6 months, the five patients show an ability to move; however, I am not sure against what baseline.
Based on a clinical score, the patients either were stable with a few showing a modest improvement.
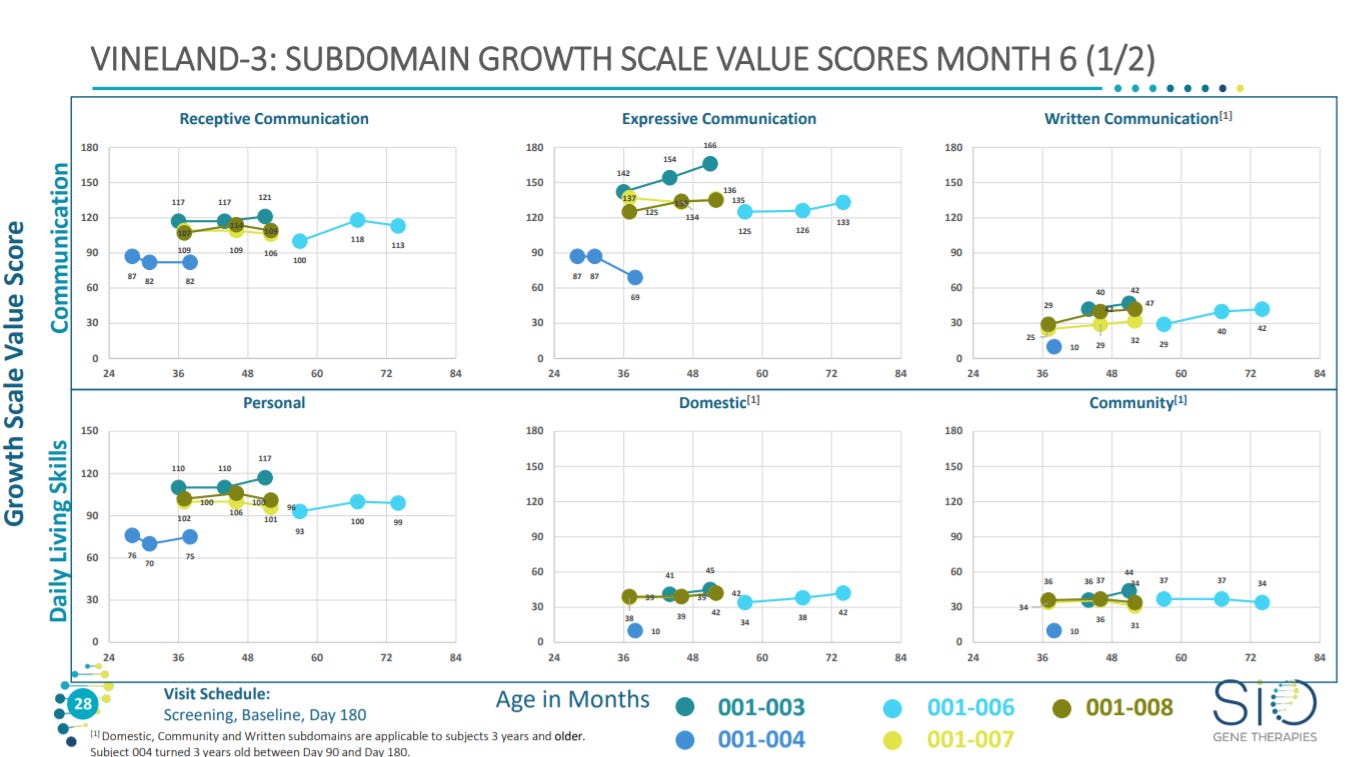
With a slide for an overview on Vineland-3 scoring to measure behavioral and intellectual disabilities - https://www.sralab.org/rehabilitation-measures/vineland-adaptive-behavior-scales#:~:text=The%20Vineland%20Adaptive%20Behavior%20Scales,%2C%20autism%2C%20and%20developmental%20delays.
Based on Vineland-3 scoring, the treatment shows no obvious significant increases. I am not too familiar with this scoring system.
More data on Vineland-3 scoring.
This slide gives an overview on the current clinical data for Sio’s lead program.
With an overview again on the lead asset again.
Sio then goes into their second asset focused on Tay-Sachs. The gene therapy dosed their first patient in early 20201 using an AAVrh8 vector to deliver functional HEXA/B genes to the central nervous system.
The natural history shows a loss of various motor skills within the first years of life.
In cat models, Sio’s drug candidate showed an ability to increase survival and show distribution within the brain.
So the clinical trial design is a dose escalation study delivered through the CSF.
Sio ends their discussion on their GM1/2 programs by touching up their manufacturing partnerships. The process is the product.
The next section goes into Sio’s program in Parkinson’s Disease.
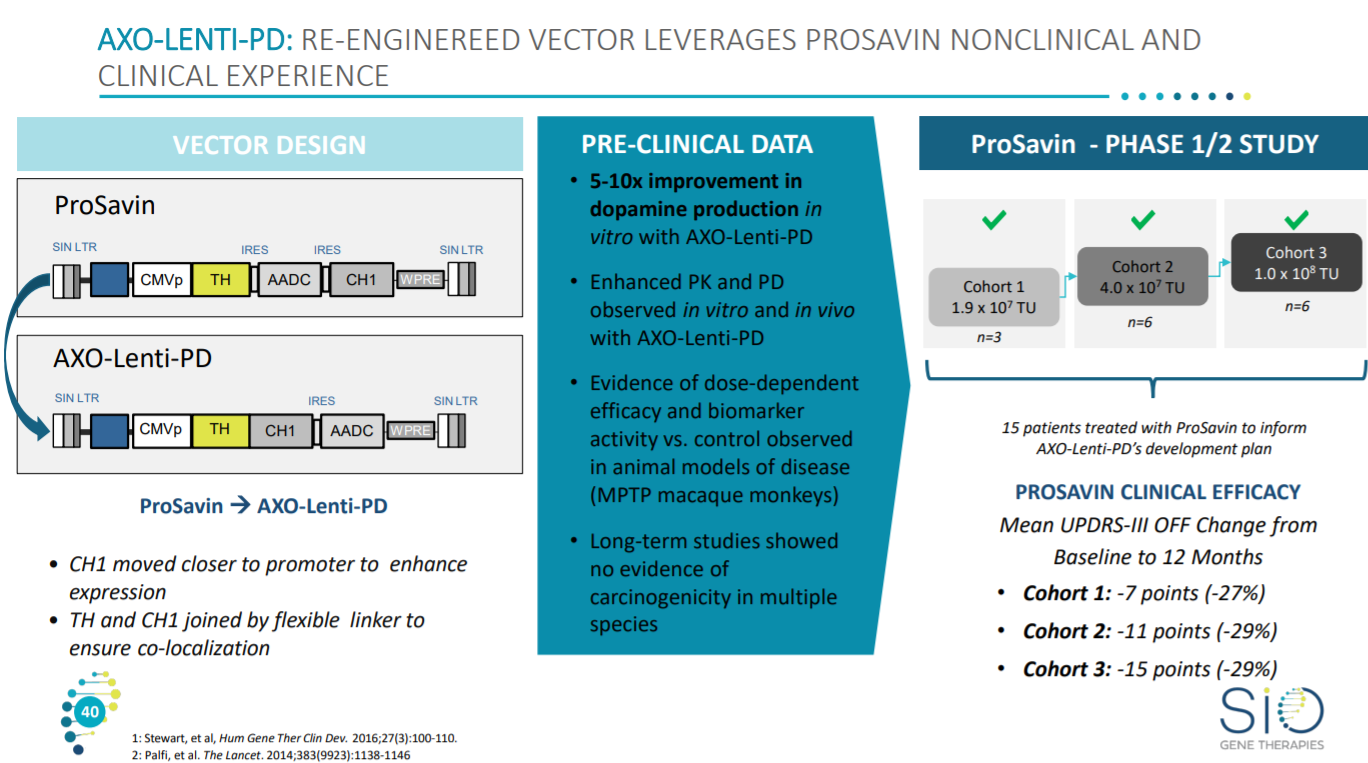
The lead asset is a lentiviral constructs to deliver 3 genes to increase production of dopamine in patients - https://www.oxb.com/pipeline-items/axo-lenti-pd-sio-gene-therapies
This slide gives an overview on the premise of the program - lentiviruses have a ~3x higher packaging size versus AAV but have safety issues from intesertional mutagenesis.
Sio’s gene therapy builds on ProSavin - https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6157351/
With the clinical trial currently enrolling for the next dose cohort.
With a slide on the endpoints.
Current clinical data (n=6) show a modest increase in these endpoints.
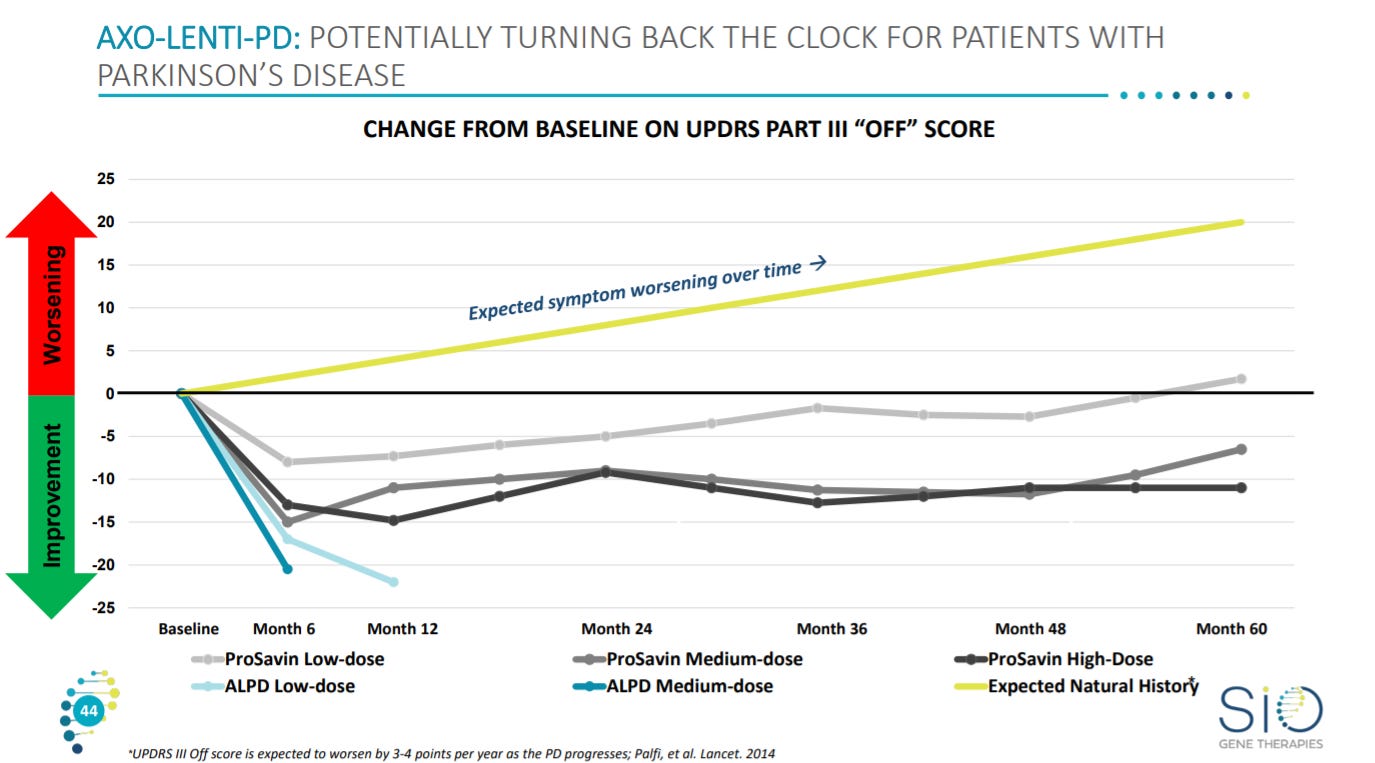
With an improvement for patients based on a Parkinson’s rating score - https://www.movementdisorders.org/MDS/MDS-Rating-Scales/MDS-Unified-Parkinsons-Disease-Rating-Scale-MDS-UPDRS.htm where Sio’s asset shows a match with ProSavin so far.
With an overview on the clinical data.
With a plan to expand to the highest dose cohort.
Along with a slide on Sio arguing the advantages of their lentiviral gene therapy for Parkinson’s versus other potential treatments.
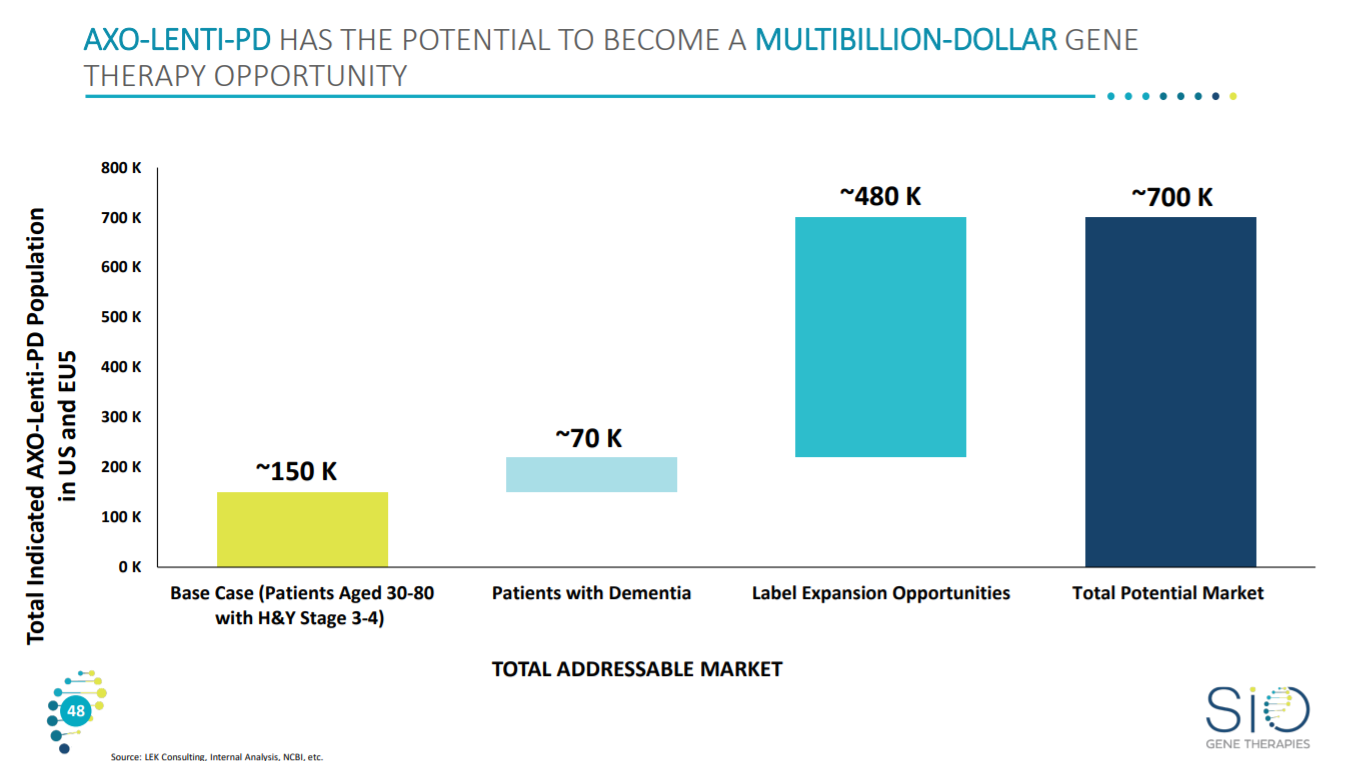
With the addressable patients being around 150K to up to 700K.
The last slide gives an overview on Sio: their 3 gene therapy programs and $88M in cash.
Sio Gene Therapies’ presentation does a good job at explaining their clinical programs, biological hypothesis, and current data. Sio has a long way to go and will need to raise more capital to bring these drugs to pivotal studies. But the company has done a great job pivoting from Alzheimer’s to building an exciting pipeline for GM1/2 and Parkinson’s.
Follow up questions for the team:
Are there any partnership opportunities for the Parkinson’s program?
What are the total costs to bring AXO-AAV-GM1 to a pivotal study?
Is Sio going to build out internal manufacturing capacities or rely on CDMOs?